 From our South American Correspondent…
From our South American Correspondent…
The Argentine national flag is from a design by General Belgrano who died on June 20 1820 – 200 years almost to the day before Fernando Polack’s strange plasma trial began.

Fernando Pedro Polack MD in the spotlight
Strange things have been going on in the murky world of clinical trials in the democratic nation of Argentina. Argentina ranks 78 out of 180 countries on Transparency International’s Corruption Perceptions Index.
There was a scandal last year when it emerged that 70 VIPs received their Sputnik V vaccinations ahead of others at the Posadas Hospital in Buenos Aires. The Ukraine conflict has now shone a further light on Russia’s vaccine diplomacy.
In a masterpiece of bad timing, President Alberto Fernández had an audience with Putin at the Kremlin three weeks before Russia invaded Ukraine. No long table in sight here.

At a press briefing on the day before they both left for the opening of the Winter Olympics, Fernandez stated:
Argentina owes a debt of gratitude… Russia was the first country to make sure that Argentina received the coronavirus vaccine… which helped us mitigate the impact of the pandemic on our people. Based on this, we can expand our technology cooperation. Together, we can launch the production of not only vaccines, but other products as well.
This was shortly after a trial suggesting the Russian Embassy in Buenos Aires was used as a conduit for supplying cocaine to Moscow – and a year after Argentina’s triumphant role in Pfizer’s American vaccine.
Famous Argentinian Doctors
Argentina has a fairly sophisticated Health Service. Childhood immunisation is mandatory although uptake is slightly behind some other nations. 48,000 doctors are concentrated in the city of Buenos Aires, making a density of 16.6 doctors per 1000 inhabitants.
Arguably the most famous Argentinian doctor was René Gerónimo Favaloro, who performed the first documented coronary artery bypass graft in 1967. He also set up a charitable medical foundation in Argentina but, in July 2000, at age 77, committed suicide, fed up with the continuing
“…fight with the prevailing corruption in medicine … We have consistently refused to break ethical guidelines; as a consequence, we never gave a single peso in return.”
Have things changed in the last 20 years?
Dr Fernando Pedro Polack heads up the list of 29 authors of the NEJMgate report of the Phase III trial of Pfizer-BioNTech’s Covid vaccine, called BNT162b2 or COMIRNATY – but more usually just “Pfizer”.
Born on the 6th March 1967 into a medical family, Fernando studied medicine in Argentina before completing his paediatric training in USA. It’s a long story trying to unravel his transition from an enthusiastic research paediatrician to an empresario who rubs shoulders with Buenos Aires glitterati. It may also seem a bit odd that a paediatrician finds himself running covid trials in adults. Fernando gives a TedX talk HERE the site also has some history on him.
Funny smell in Buenos Aires?
Buenos Aires sits on the west bank of Río de la Plata – literally “River of Silver”. Argent also means silver – the silver natives gave to Europeans who landed there.
Fernando has been commercially involved in clinical trials since 2014 when he was the director of Unitrials SA in 2014. This changed its name to Infant Trials SA in 2015.
iTRIALS SA was formed the following year. His co-directors are biochemist Silvina Andrea Coviello and Fernando’s guitarist son Leandro. There are now four iTRIALS subsidiary LLC companies registered in Florida, c/o the offices of Cervetta-Lapham & Associates.
iTRIALS is a site management organisation (SMO).
“iTRIALS assists sponsors, CROs [Contract Research Organisations] and investigators in all aspects of clinical trials to help ensure that goals and expectations are timely and successfully met with international quality standards.”
“iTRIALS identifies high volume clinical research sites of extraordinary recruitment potential and high-quality staff.”
iTRIALS shares its offices with another of Fernando’s babies – Fundación INFANT. The combined HQ (below with the black entrance) is conveniently located next door to Freddo, an ice-cream parlour.

Freddo Ice-cream parlour, next-door to iTrial’s HQ at Gavilán 15
When Covid struck Argentina, Fernando and his Fundación became involved in a trial of immune plasma, taken from patients who had recovered from Covid, given to patients who had recently acquired the disease. In May 2020 he speculated that this would make Covid like an ordinary cold, and the Gates foundation would offer financial support. He used high profile press conferences to disseminate his exciting message.

Polack at his Plasma press conference

The gang of five that ran the plasma trial at the Hospital Militar
This trial was run by the same people from June to October 2020 – overlapping almost exactly with the Pfizer trial. It claimed a positive outcome. The results were published in a peer-reviewed NEJM article in Feb 2021.
The conclusion in the abstract bizarrely states:
Funded by the Bill and Melinda Gates Foundation and the Fundación INFANT Pandemic Fund; Dirección de Sangre y Medicina Transfusional del Ministerio de Salud number, PAEPCC19, Plataforma de Registro Informatizado de Investigaciones en Salud number, 1421, and ClinicalTrials.gov number, NCT04479163.
Is this the first time the conclusion of an abstract of a randomized controlled trial in a medical journal is given over almost entirely to a statement about the funders?
A subsequent systematic review and meta-analysis failed to confirm these findings, noting “very serious imprecision concerns”.

Meanwhile, on 10 July 2020, without any prior announcement, Fernando Polack and Nicolás Vaquer, CEO of Pfizer in Argentina, called on President Fernandez to tell him that Argentina had been selected by Pfizer to conduct Phase III clinical trials of their Covid Vaccine. Polack had taken part in Zoom calls with Pfizer since May that year.
Infobae reported Fernando proudly saying
“…if you have the scientific privilege of participating in the evaluation of a vaccine, you contribute to potentially better position your country in the waiting line for distribution.”
This placebo-controlled trial was arranged with military precision. Fleets of taxis were booked to ferry volunteers between their homes and the vaccine clinic at Hospital Militar Central in the northeast of the city. Volunteers were each given a tablet with a pre-installed app to record pre-defined symptoms.
About 5,800 volunteers were enrolled, half getting the active vaccine. This is almost 4 times more than the next largest centre in this trial. Amazingly 467 doctors were almost instantly signed up and trained as assistant investigators in the study. Fernando was in command as Pfizer’s Principal Investigator.
The first article about the Pfizer-sponsored multinational ground-breaking study was published in the New England Journal of Medicine on 31 December 2020, with Fernando as first author. Is this the first paper in NEJM with the owner of an SMO as first author?
The second author was Dr Stephen Thomas of the State University of New York, Upstate Medical University, Syracuse. In a BMJ podcast, Dr Thomas bills himself as the Lead Principal Investigator. His academic centre enrolled 364 volunteers.
Despite leading this project, Dr Thomas makes it very clear that he was only allowed access to analyses of aggregated data and didn’t know when or if Pfizer would release the raw data. It is not clear how much or how Pfizer paid for SUNY participation in the trial.
Our question is: Who ran the Buenos Aires vaccine trial, and where did the money go?
Was it Dr Gonzalo Pérez Marc from the Hospital Militar Central (where Augusto Roux was vaccinated)?
Several press reports claim that he coordinated the trial. He was a co-author on the NEJMgate article but is listed by Pfizer only as a Sub Investigator in the Covid trial. He was previously employed as Principal Investigator in Pfizer studies of RSV in pregnant women. Until last year Gonzalo was also Chief Operating Officer of iTRIALS.
Was it Fernando?
He is the first author of the first NEJM article and Pfizer documents show him to be the Principal Investigator in Argentina. But he says his role in the trial was merely as an associate investigator at Departamento Materno Infantil in the Hospital Militar.
All NEJMgate authors have to submit a “Disclosure Statement” to show any potential conflicts of interests.
Polack reported grants from Novavax and personal fees from Janssen, Bavarian Nordic A/S, Pfizer, Sanofi, Regeneron, Merck, Medimmune, Vir Bio, Ark Bio, Daiichi Sankyo outside the submitted work.
At least eight of these companies are engaged in RSV vaccine research in babies and pregnant women. Fernando has mentioned a combined RSV, flu and Covid vaccine.
He also disclosed a grant from “Pfizer as investigator and site management organization co-owner” (iTRIALS).
Here is the problem. As President and shareholder in iTRIALS why did he (and Marc) claim “that they have no financial interests in this product”?
Enter Baker McKenzie, international lawyers with an office in Buenos Aires.
It is claimed that they brokered a joint venture between iTRIALS and Pfizer for the Covid-19 vaccine clinical testing programme
No one knows how much Pfizer money was channelled into iTRIALS. Small family doctor operations in Nebraska with no prior research experience have registered several million dollars in income for this period.
Pearly Gates
Fernando also doesn’t mention his extensive financial involvement with the Bill & Melinda Gates Foundation. This organisation supports industry vaccine trials including Covid and RSV. Fernando is heavily involved through his Gates-sponsored Fundación INFANT in Buenos Aires in RSV trials and research.
Gates sunk $82,553,834 into Novavax’s RSV vaccine ResVax which was shown to be ineffective in clinical trials in pregnant women.
Pearl River in New York is the site of Pfizer’s Covid vaccine efforts. It is also the site where, having been frozen, all blood and nasal swabs from Argentina were sent and are stored.
Argy-bargy
Who knows what the roles of Polack, iTRIALS and his various foundations have been in both clinical research and his dealings with global pharmaceutical companies.
We do know something more definite about the ethical responsibility that goes with these trials and his dealings with some of the patients.
Disappeared in Argentina tells the tale of Augusto Roux, volunteer number 12312982 in Pfizer’s Buenos Aires Covid Vaccine trial. Dr Polack was Principal Investigator.
To recap – Augusto received his second injection on 9 Sep 2020. He did not know whether this was the Covid vaccine or placebo. On the Sep 12 he was admitted to Hospital Alemán with a fever and vomiting. He was fully investigated, including a CAT scan of his chest. This (attached) showed an unequivocal pericardial effusion. That is an abnormal collection of fluid around the outside of the heart. On 14, Dr Ivonne Name, one of the investigators, coded this as a severe reaction to the vaccine.
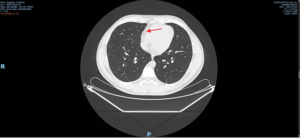
This is the report of his CT scan of the chest from Hospital Alleman on 12th September 2020.
Pleura: Dentro de límites normales.
Vasos: Dentro de límites normales
Corazón: De tamaño normal con derrame laminar pericárdico.
Mediastino e hilios: Libre de adenomegalias.
“Derrame laminar pericárdico” is translated as “laminar pericardial effusion”
Remember this was in the early days of Covid Vaccine trials. Investigators, like Polack, should have been knocking on the door of Hospital Alemán trying to find as much information as possible about this adverse event. Augusto gave them permission to do so. Instead on 17 September Polack writes in Augusto’s record:
 the hospitalisation was not related to the vaccine.
the hospitalisation was not related to the vaccine.
Furthermore, he later diagnosed Augusto as suffering from anxiety when he tried to find out whether he, Augusto, was in the vaccine or placebo group.
Neither Augusto’s pericardial effusion, nor another volunteer’s penile vein thrombosis, appear to have found their way into the reported side effects of this trial.
Altogether close to 20% of the patients in this trial were recruited by i-Trials or Ventavia and likely over 90% by commercial entities – see Eric Rubin.
DH Comment:
Back to Earth
In early summer 2020, the Lancet and the New England Journal of Medicine published—and rapidly retracted—major covid-19 studies based on fraudulent datasets.
Pre-empting Pfizer, in August 2020 Vladimir Putin claimed the Sputnik vaccine works. In September 2020, the Lancet reported two small open-label studies supporting this claim. They generated scorn and disbelief that the Lancet would publish them – Van Tulleken.
On February 2, 2021, a Sputnik phase III study was reported – again in the Lancet – claiming over 90% effectiveness. The academic view was
The overall impression is that of inadequate peer review and editorial processes that failed to detect what seem to be obvious errors in reporting the results of this high profile research
The Lancet’s website makes it clear that, like many journals, it does not have access to “raw data related to research studies”.
On February 2 2022, the day Putin and Fernandez met, the Argentine regulator approved Sputnik, previously used widely under emergency authorization. Just over three weeks later, after war in Ukraine broke out, the US and Europe sanctioned the Russian Development Investment Fund, the sponsors of Sputnik.
Sputnik seems unlikely now to get approved by Western regulators (Tienari), even though six weeks after the Putin Fernandez meeting a Lancet paper from Argentina claimed that Sputnik and the Astra-Zeneca vaccine have over 90% effectiveness in real world settings – reducing deaths and hospitalizations etc.
Not to be outdone by the NEJM, the Lancet organized for an editorial praising this real world data, authored by a CRO operator, given an academic link by McMaster University, who ironically claim to be the home of Evidence Based Medicine (EBM). Its perhaps not surprising that what our new cadre medical ‘academic’ businessmen say should follow the money – in would infringe business ethics not to.
Notwithstanding an input from the ‘home’ of EBM, observational, real world, data like this are usually held at arms-length by most medical academics who sniffily note these data are not from Randomized Controlled Trials.
The problem for NEJM medical academics is that the Pfizer RCT, even with the best input from commercial SMOs in the US and Argentina, shows negative effectiveness for Comirnaty – more people died on the vaccine than on placebo.
Confronted with these RCT data, academics and the commentariat have turned to unpublished real-world data. There are, they say, much fewer vaccinated people dying or being hospitalized compared with the unvaccinated. There isn’t even a fig-leaf of peer review for these real world data, which are taken from the dashboards of public health departments in the business of enforcing vaccine mandates.
Forty years ago, companies began denying access to trial data and ghostwriting journal articles. Thirty years ago, when faced with serious harms on Prozac, Eli Lilly, its makers, hid an excess of suicidal events (including death) on Prozac in a BMJ article.
With this article, Lilly were the real creators of Evidence Based Medicine in the sense of the people who persuaded medics to adopt this approach rather than the first person to adopt the slogan. This article opened the door to a state of affairs where the greatest concentration of fake literature on earth now centers on the drugs that doctors give patients, with the proportion of that literature that is fake now close to total.
Chris Van Tulleken is a decent man as are the current editors of the BMJ. In his BMJ review of the Sputnik saga he said: ‘Journals risk being used in place of regulators when they publish studies of novel vaccines … peer review is inadequate to decide the risk-benefit ratio of new drugs’.
The … is the spot where Chris suggests that review by regulators is more robust than peer review because:
- Regulators see the data – he and BMJ’s editors know they don’t.
- Regulators don’t have to cope with the Sally Ladens of this world who ghostwrite journal articles – they do; the Sallys write the Clinical Study Reports also, increasingly with the help of Artificial Intelligence (A.I.), probably soon to be the other way around.
- FDA will not approve treatments based on negative studies – Chris and the BMJ editors know they do as in the approval of Prozac and Paxil for children.
- FDA would not allow negative studies to be published as positive in major journals like NEJM or the Lancet – Chris and BMJ’s editors know they do and that, as Erick Turner has shown, a large proportion of antidepressant trials are published as positive in major journals that companies have told regulators are negative.
One solution to this problem lies in access to the data by a wider public – this does not mean ‘responsible access’, nor does it mean which medics have had what monies from pharma. Without unfettered access to the data, the Pfizer Science will Win slogan is risible. Its a gag both in the sense of a joke as well as something put across the mouths of people to stop them speaking.

Another solution is to recognize the harms that drugs and vaccines are causing and stop gaslighting people like Bri Dressen in the US, and Augusto Roux in Argentina, by saying reports of harms are anecdotal, misinformation, lacking in scientific support. For a doctor to do this to a patient is unconscionable.
Lawyers attempting to cleanse the trial record of evidence of harms comes very close to fraud on whatever legal system might ultimately have to deal with these harms – see Eric Rubin Boston Strangler.
When it comes to vaccine harms, Western governments are being staggeringly autocratic (in the sense of driven by algorithm) – see One Algorithm to Rule Them All.
They are steadfastly adopting the role of a Bad Samaritan. At present only one in 535 Members of Congress is speaking up about harms. If the Duma has even one in its 450 Members speaking up about treatment related harms, it will demonstrating greater decency and humanity (in this respect, whatever about others) than Congress.
As outlined in the Cause and Effect Forum, we have reached a point in respect of medical treatments where it is only in considering the harms of treatment that we can do proper science, in the sense of working on problems which we can settle with data.
It is only when interviewing a harmed person that the data we need is fully present. Science can then get done – if a doctor or other healthcare person is alone in the room with the harmed person, without the strangers who set guidelines and mandates and threaten to revoke her or his medical license, whose presence is all too real and far from ghostly for the person who seeks recognition and help for a harm.
Faced with cases like Karunya’s, Nina’s and others to come in the Forum, we can arrive at judgments that are a best possible fit with the data, in a way that we no longer can with any of the other data swirling about (except some of the biological data). Those who are harmed are the Cracks through which the Science gets in.
Updates
Meanwhile the NEJM yesterday, March 23, features Pfizer’s booster trial – which appears to have Ventavia in and iTrials out, even though by a long long way the largest number of boostable trial participants from one center came from Buenos Aires.
In the Lancet on March 24, there is an article supported by the National Key R and D Program of China, which reviews vaccine trials data availability concluding that:
The reporting and data sharing level of COVID-19 vaccine trials were not optimal.
We have been asked how much Nebraska docs earned from this trial – Eric Rubin. Here is a table with Platinum Research Network Doc’s earnings.
| Name | City | Doc | Money |
|---|---|---|---|
| Tekton | Austin | Pickrell | $3.5 M in 2020 |
| Ventavia | Houston | Van Tran | $2.9 M in 2020 |
| Ventavia | Keller | Fuller | $2.9 M - mostly 2020 |
| Ventavia | Fort Wort | Koch | $1.6 M in 2020 |
| Sterling | Cincinnati | Butcher | $1.2 M 2020 |
| Sterling | Cincinnati | Davit | $ 1.0 M 2020 |
| Meridian | Savannah | Bradley | $2.9 M 2020 |
| Meridian | Omaha | Essink | $4.9 M 2020 |
| Meridian | Norfolk | Harper | $1.8 M 2020 |
| Meridian | Sioux City | Ensz | $1.8 M 2020 |
| Meridian | Binghampton | Eder | $1.6 M 2020 |
| Meridian | Endwell | Suchet | $2.6 M 2020 |
| Sundance | St Louis | Wadsworth | $1.9 M 2020 |
| Medpharmics | Gulfport | Matherne | $3.1 M 2020 |
If the same rates of pay held per patient recruited for Fernando and colleagues, they took in well over USD $50 M – given their role as owners of the Buenos Aires’ equivalent of Ventavia in addition to their medical roles.
¿Por qué?
And as for fortune, and as for fame
I never invited them in
Though it seemed to the world they were all I desired
They are illusions
They’re not the solutions they promised to be
From Don’t Cry for Me, Argentina.
Composed by Andrew Lloyd Webber and Tim Rice.

The running Brook…
Brook Jackson
@IamBrookJackson
SASHA LATYPOVA’S FORENSIC INVESTIGATION INTO PFIZER’S ‘FRADULENT’ PRECLINICAL STUDIES
It really is shocking stuff…
https://www.dropbox.com/s/gh08pl6sp9w1o11/GMT20220321-170215_Recording_3240x2160%281%29.mp4?dl=0
“You can’t have multiple versions of it, it is just fraud”
“You can speed up only fraud”
“The public was sold this fairy tale”
“Pfizer Cite – WHO” …
Argentina’s health minister fired in ‘VIP vaccines’ scandal
https://www.ft.com/content/23828ca6-b684-4dca-9efa-d72765d52bb4
An American, a Brasilian and an Argentinian go take a test together
In it, they all have to get in a plane, take off, go to random location, and just by putting their hand out of the window mid flight, they have to guess if they are in their countries or not and why.
First goes the american, who says:
– We are in the USA, i can feel the freedom of democracy just by feeling the air.
Second goes the Brasilian who says:
– We are definitely on brazil, i can feel the cultures, the happiness and samba in the wind-
Lastly goes the argentinian who says:
– Nope, we are totally on Argentina, no doubts about it-
– Why are you so sure?- asks the interviewer, to which he responds:
– Because someone stole my watch-
WATCHgate NEJMgate…
So how did Argentina find itself relying on Russia’s Sputnik vaccine in the first place? Why not go with Pfizer? According to reports that emerged a year later, Pfizer engaged in “high-level bullying” of Latin American countries in 2020 negotiations over vaccine access. As a result, both Argentina and Brazil walked away without a deal as the pandemic raged.
The key sticking point? Immunity from lawsuits over vaccine-linked injuries.
Now, other Covid-19 vaccine makers did ask countries to “indemnify” them against vaccine injuries, and compensate their own citizens for adverse effects. And most nations agreed – based on the idea that some injuries would be 1) inevitable but 2) very rare and 3) mostly not serious or fatal. But Pfizer went much further. They also insisted on immunity from suits alleging negligence or misconduct by Pfizer itself – and demanded that key national assets, from embassies to military bases, be signed over as collateral to back up this pledge!
In other words: Despite Pfizer’s appetite for Argentine bodies to test their novel vaccine in, they felt no duty to provide the actual product once it came to market. It’s a familiar and ugly state of affairs that’s all too common in Pfizer’s home country: New drugs are tested on poor folks who (assuming the drugs work out) may never get access to them.
Not for the faint-hearted
UK government “yellow card” adverse reaction reports
https://www.gov.uk/government/publications/coronavirus-covid-19-vaccine-adverse-reactions/coronavirus-vaccine-summary-of-yellow-card-reporting (25 Mar 2022).
“Up to and including 9 March 2022, we have received 746 reports of myocarditis and 514 reports of pericarditis following use of the COVID-19 Pfizer/BioNTech Vaccine….Four fatal events have been reported associated with the COVID-19 Pfizer/BioNTech Vaccine.”
Pfizer should track down and invite these people to have periodic independent cardiological assessments over the next 10-20 years.
They should also channel 0.1% of their profits into research into why a mRNA “vaccine” should cause heart problems.
Pfizer deserves a red card.
Evaluating COVID-19 vaccines in the real world
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(22)00194-5/fulltext?dgcid=raven_jbs_etoc_email
The effectiveness of the mRNA vaccines in preventing COVID-19 disease progression in 2021 set new expectations about the role of prevention interventions for the disease. Efficacy observed in the trials was more than 90%.
The efficacy of other vaccines evaluated in large randomised trials, such as the Oxford–AstraZeneca (70%) and Sputnik V (91%) vaccines, have been criticised for elements of the trial conduct and concerns about safety.
For that reason, mRNA vaccines have been most widely distributed
EJM is employed by Platform Life Sciences, which is a private company providing consultancy on global health trial design and methods. Platform Life Sciences is not involved with any work on COVID-19 vaccines. EJM has previously been employed by Cytel, which does contract research and software development. GR is the founder and an employee of Cardresearch, which leads the Brazilian TOGETHER trial of repurposed therapies for SARS-CoV-2.
References
Baden LR, El Sahly HM, Essink B, et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N Engl J Med 384: 403–16.
Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med 383: 2603–15.
Juan Gérvas Retweeted
@samizdathealth
@DrDavidHealy
Did the New Eng J Medicine sell itself down the River of Silver? Can anything it has published on the vaccines be trusted when its editors don’t see it as their job to guarantee the integrity of their sources?
@JeanRees10 @DrNancyOlivieri @guyattGH @eloder
As a radiologist, I can confidently say that there is no pericardial effusion on that chest CT image (despite using a lung window and not soft tissue window). Just minimal pericardial fluid, which is normal.
Everything else is outside my purview!
Difficult for me to argue against a radiologist
except to say the radiologist in the hospital where A was treated note a pericardial effusion
and I had the full sequence of X Rays reviewed by a radiologist who knew nothing of the story who diagnosed a pericardial effusion.
But the pericardial effusion is not all that’s involved, there are the liver enzymes and a concerted effort by the research team to give Augusto illness he didn’t have – I imagine even a radiologist would not put much weight on a mental health diagnosis made by a pediatric infectologist who had a vested interest in discrediting this trial volunteer.
D
because in my country who can perform an imaging diagnosis through a computed tomography is a doctor with that specialty, the radiologist only takes x-rays. That is why there is a code of ethics in medicine and I fully understand that you cannot observe it, each one with their qualifying title, greetings from Argentina
Lcdo:
Mil gracias por compartir con todos su experiencia. Lamento mucho los malos ratos que ha pasado no solo por los efectos adversos suscitados por las inyecciones sino tambien por los obstaculos y maltratos que ha tenido que encarar. Ojala recupere plenamente su salud, que sea muy pronto.
Saludos
M Regan