I was invited by Yulia Mikhailova to deliver a Guest Lecture to the New Mexico Technical Institute on Wednesday February 2. The talk began and appeared to be going smoothly, when halfway through the Zoom room was hacked with the hackers saying and writing a lot divisive and hateful things. A later post will say more. The talk has been re-recorded and hopefully a recorded Q and A will follow soon.
The recorded talk Who’s Afraid of Science is here and the slides and text are below for a talk that offers the message that science, democracy and love are intimately interlinked .
WHO’S AFRAID OF SCIENCE?
It’s a great pleasure to be speaking to you here today. I hope at the end of this talk you will realize that really doing science is scary but I also hope your fear will lead you to want to do it even more.
Slide 2
In 1649 England had a World Turned Upside Down – a Storming the Capitol – moment. The King, Charles I, had his head chopped off. Not by an invading army. Not by a usurper to the throne but by his subjects who wanted a different form of government.
Slide 3
In 1660, the monarchy was restored. Charles II brought something new into being – The Royal Society – an initiative that has played a huge part in how we are governed now.
The Royal Society established the ground rules of Science. It would appeal to the data people could see in front of them. Whether you were Xtian, Hindu or Muslim, Jew or Atheist, you left these badges at the door and had to explain what you saw in front of you without appealing to a Book or Authority. You were encouraged to run the experiment again, adapt the apparatus, or make sure there was no trick – what you couldn’t do was refuse to come to a conclusion.
Slide 4
We view going by the data now as central to science and who we are. But Scientists overlook an equally important event 44 years’ earlier, when in 1618 Walter Raleigh had his head chopped off.
Raleigh’s execution was not an experiment to see if we could live without heads. He had been convicted on the basis of people who didn’t like his politics saying things about him which were used in evidence against him – but crucially these people did not come into court to be cross-examined.
Legal systems worldwide recognized the injustice of this and introduced Rules of Evidence. Hearsay could not be used as evidence. The only evidence that can be used in court is material that can be put in front of jurors who can see the people or material being examined and cross-examined.
The process of forcing 12 people with very different biases to come to a verdict about what is in front of them is the very essence of science.
A verdict is obviously provisional – this is the view that best fits the facts we were presented with but clearly if the facts change then we reserve the right to change our mind. This might appear to contrast with what we now think of as the objectivity of science, but scientific views are similarly provisional with participants in the scientific process encouraged to attempt to overturn the verdict with replicable data.
Just like the scientific approach lands us on the moon and helps us explode atomic bombs, so the judicial process has to be rigorous because ultimately on the basis of these verdicts we execute people. I am not talking about humanities here or social science. I am talking specific verdicts that bring life, death, or incarceration in their wake.
Slide 5

Do you think studying monolayers of cells will tell you
why you fall in love with a girl?
I began in Neuroscience when the major figures had been trained by people like Walter Hess, who won the Nobel Prize for Medicine in 1949 – people who knew that whatever you found out about cells or neurotransmitters everything was ultimately about function. Life and death.
And of course, I was desperately trying to understand girls and how to get them to fall in love with me.
Slide 6
Serotonin was just coming into the frame when I started and I began looking at serotonin reuptake in people who were depressed – before we had SSRIs – triggered by this Jouko Tuomisto article here.
Doing serotonin reuptake and related assays gave me a feel for the importance of methods and how little things can change an interpretation. Toumisto found lower serotonin reuptake in depression. I found that it depended on the time of day you ran the assay. If you did more than one assay per day the picture looked quite different. Laboratory data usually raises more questions than provides answers.
Work on serotonin introduced me to the idea of biobabble. When the SSRIs came on the market it was clear the talk about chemical imbalances and low serotonin was just marketing copy. It had and has no scientific meaning
Slide 7
My lab research gave me the credentials to interview the grandees in psychopharmacology. I could ask questions that let people from the lab, the clinic and industry feel able to open up.
Here you see Arvid Carlsson who won the Nobel Prize for discovering the role of dopamine. He also made the first SSRI – zimelidine. He was one of 100 psychopharmacologists interviewed.
Companies came my way because I looked like just the kind of person to educate doctors about the new drugs. Industry people were the most candid about these new drugs not being as effective as the old ones but pretty confident they could market them. Doctors were also relaxed with me – I was one of them. Everyone opened up.
Slide 8
The Dutch here translates as Nothing is out of Hand – that is Everything is in Control.
Two men who came into my life in 1990 shaped it from there on. When the SSRIs came onstream I used them before my peers interested to get a sense for how they worked. A senior university academic wanted to be less obsessive which SSRIs can help with. So, we tried the just released Prozac. He came back intensely agitated and thinking about killing himself. We stopped Prozac – the problem cleared up. We waited a month and then I mentioned there was another drug that was good for OCD we could try. We tried it and all the suicidal feelings came flooding back. We stopped and they cleared up again.
A just retired senior executive had been put on Prozac and ended up with me because he’d gone to a local quarry to drown himself, changed his mind and then tried to walk out into the sea but it’s difficult to drown yourself off the North Wales coast – the sea is too shallow. The Prozac was stopped and he improved but confessed to other thoughts about killing himself.
I put him on an older antidepressant, but also a serotonin reuptake inhibitor, and could see him become visibly more tense and agitated over a few days. We stopped the drug and he was fine again.
Slide 9
I wrote these two cases up in the article you see here and presented them at several meetings. There was no other way to explain what had happened, other than the SSRI did it. Both men when challenged with the drug had problem that cleared when the drug was stopped and reappeared when a similar drug was introduced. I sent a draft to Lilly, who make Prozac, asking if they had any thoughts on what was going on. They didn’t, which seemed odd.
Slide 10
At the same time as my article came out, Lilly published this article by Charles Beasley et al. It came out in the BMJ on the same day Lilly presented the case in the article at an FDA hearing September 20, 1991 – just over 30 years ago.
From a year before, there had been convincing reports of people given Prozac, becoming suicidal, with the problem clearing when Prozac was stopped, and reappearing when it was restarted. In terms of cause and effect, there was no doubt the drug caused the problem – it would be like reintroducing oxygen into the air pump and seeing a dying bird come back to life.
FDA were forced to have a hearing on antidepressants and suicide, at which Lilly pitched their RCTs stating:
- the plural of anecdote is not data
- it’s the disease not the drug
- are you going to believe the anecdotes or the science?
The Beasley article shows more suicidal events on Prozac, but the paper said these were not statistically significant and BMJ were happy with a statement that the problem didn’t exist.
FDA talked about heart-breaking cases reported to them but concluded the science didn’t support the drug causing the problem.
Slide 11
It’s common to hear distinctions between the Art and the Science of Medicine. This is caught in this famous painting of the good doctor – who is not doing anything but waiting with the sick child and their family. But of course, once he or she could do something waiting without doing it would not be a good idea – none of us would go to a doctor like this.
Although as Philippe Pinel said, there is an art in being able to do something in medicine but an even greater in knowing when not to do it.
Slide 12
Those in the social or human sciences, soft scientists, talk about the richness of the qualitative context compared with the experimental context of the hard sciences, where hard scientists want to eliminate confounders. But this pre-Risperdal Janssen Advert assumes no one in medicine pays much heed to that.
Slide 13
But we need to avoid confusing controlling confounders with standardization. Standardization can help us control confounders but standardization without judgement calls gives us Big Mac hamburgers rather than really good hamburgers.
At some point the need for judgements, verdicts – diagnoses comes into the frame.
The first rating scale for behaviour was the Hamilton Rating Scale for Depression. Here is Hamilton saying:
It may be that we are witnessing a change as revolutionary as was the introduction of standardization and mass production in manufacture. Both have their positive and negative sides
He saw the scale as an aid to make sure physicians checked on most of things that might be abnormal in depression while interviewing a patient. An aid to help a doctor do a good interview.
Slide 14
An aid to help you live the life you want to live or to get you to live the life Pfizer want you to live?
If you cleave to the checklist you will do very standardized but possibly disastrous interviews. For instance, on the Hamilton Scale, there is an item on suicide which could stem from the illness or from the drug. This needs a judgement call – if caused by the drug you should rate the person as Zero – if caused by the illness you might rate 3 or 4. Ditto for sex, for sleep. Just checking yes for suicidality risks going badly wrong.
Checklists like these however became viewed as scientific instruments. They look better to hospital managers than David Healy asking you about the football last night or your family.
For drug companies, rating scales ensure you do an interview that produces figures which are the most seductive way to get people on their drug. The interview helps you to help them to live the life Pfizer want them to live.
Rating scales and operational criteria like DSM eliminate Judgement – and if you do that you are no longer doing science.
Slide 15
This is true for any measure – peak flow rates, bone densities, blood pressure or lipids, or sugar. It may be important to do something about some figures, but the goal is to help people to live the life they want to live – not getting them to live the life Pfizer want them to live.
Up until the 1980s, we brought our problems to doctors – seeking help to live the lives we wanted to live. After that they began to give us problems with out lipids and other risks and the amount of medicines being consumed rose dramatically and we increasingly began to live the lives companies wanted us to live.
A stopwatch can be a wonderful motivator to achieve a dream – breaking the 4-minute barrier for the mile. But, it just provides data from one fraction of our lives. If we remain on top of that, that’s fine – but what about weighing scales? Just after they were introduced we got the first descriptions of anorexia nervosa. In the 1920s, weighing scales had norms for ideal weight attached to them and eating disorders mushroomed. They migrated into our homes in the 1960s and eating disorders became epidemic.
It’s very difficult to ignore figures for weight. Without data from every other aspect of our lives at the same time, we risk being trapped by this one data source. We become neurotic.
Can we leave bone densities thin, peak flow rates low, lipid levels high? Yes, we can. You think of post-mortems as something that reveal what we died from – they more often reveal what we can live with.
Slide 16
Figures create risks and pharma makes money from treating risks rather than diseases. We are seduced into taking drugs when we are healthy.
The Covid dashboards are a great mechanism to generate perceptions of risk and fear. The vaccines of course treat risks – not disease.
The Meatloaf title Paradise by the Dashboard Light is what Pfizer sees but its Hell by the Dashboard Light for us – this now extends to the evaluation of lectures and ensures we pander to people rather than challenge them.
It’s extraordinary how little we have put into treating SARS-Cov, and its associated pneumonia. As Goldman-Sachs recently said Curing diseases is not a good business model.
Slide 17
Randomized Controlled Trials (RCTs) were introduced in 1962 following the thalidomide crisis. We thought they would make it harder for pharmaceutical companies to bring ineffective drugs on the market and this would make us all safer. Few notice that its mostly pharma companies who hold RCTs up as offering gold standard knowledge of what drugs do.
Here you have Tony Hill, the man who created RCTs, 20 years after the first one saying that they can be helpful for assessing one of the 100 things a drug does – something we might be able to use for treatment purposes.
This by definition means RCTs are not a good way to evaluate a drug. These days saying something like this is like telling you the Bible, the Koran or the US Constitution aren’t reliable. Tony Hill would be accused of misinformation – as would I in giving you this lecture.
Slide 18 But watch this.
Slide 19
In a depression trial, investigators focus intensely on one thing – does Prozac have an effect on mood. Pretty well everything else is ignored. The statistics we use don’t work unless there is an intense effort to collect everything we can about this one outcome.
And so, depression trials miss something that happens to almost everyone who takes an SSRI within 30 minutes of the first pill – your genitals go numb. You can search the RCTs on these drugs and all you will find is that perhaps 5% of people have sexual issues on these drugs.
If we just depend on RCTs, we end up knowing almost nothing about a drug. It gets worse.
The idea that an RCT shows there is a favourable Risk-Benefit ratio for a drug or vaccine can only hold true if the thing we are looking at is the commonest thing this vaccine does – like a parachute for instance. The commonest thing is a life saved and the Risk Benefit is favourable but we don’t need an RCT for parachutes.
Claiming a favorable Risk-Benefit ratio, if what we are hoping for is pretty rare, as in SSRI trials, and if we don’t know what we are missing, such as an obliteration of our ability to make love, perhaps for all time, is psychotic.
Slide 20
With RCTs and rating scales, we supposedly strip away context in order to control confounders.
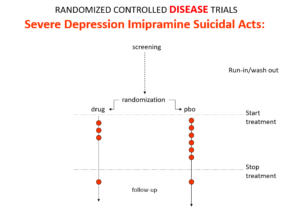
Imipramine was the first antidepressant. The older tricyclic antidepressants are stronger than SSRIs. It beats them in RCTs. It can treat melancholia – they can’t. Melancholia comes with a high risk of suicide.
Imipramine was launched in 1958. A year later at a meeting in England, Danish psychiatrists made it clear that while it was a wonderful treatment it made some people suicidal.
Let’s do a thought RCT of imipramine versus placebo in melancholia. Even though it can cause suicide, we would expect it to reduce the number of suicides because it treats the condition. This RCT would be great evidence antidepressants do not cause suicide.
Slide 21
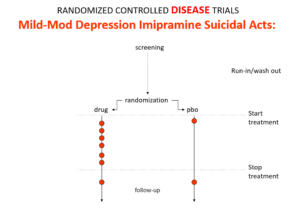
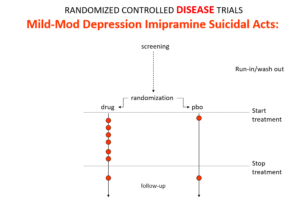
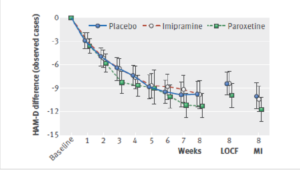
Here is the data on the trials in mild depression that brought the SSRIs and SNRIs on the market – you see a doubling of suicidal events compared to placebo. Companies resorted to all sorts of maneuvers to hide this risk.
Slide 22
This is what imipramine looks like in the same mild depressions. Now it too causes suicides. So RCTs tell us nothing about cause and effect – they can give us diametrically opposite answers. This is because these aren’t drug trials. They are Treatment Trials and in any clinical Trial, the condition confounds the effects of the drugs – and these confounders hide drug effects.
People evaluating drugs before RCTs knew this. People doing RCTs don’t. When a patient becomes suicidal in a trial you have to use your judgement to work out what is happening but RCTs, like rating scales, are all about not letting investigators use their judgment.
Slide 23
This is not just the case for depression – it’s true in every clinical situation where drugs and conditions cause superficially similar effects – diabetes and glitazones both cause heart failure, osteoporosis and bisphosphonates both cause fractures
Slide 24
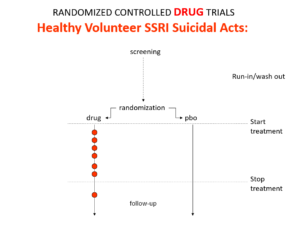
Here is what a drug trial looks like. Companies ran these studies in the 1980s and found that SSRIs make healthy volunteers suicidal, cause dependence and sexual dysfunction. These Drug Trials enabled companies to engineer their Treatment Trials to hide these problems.
Slide 25
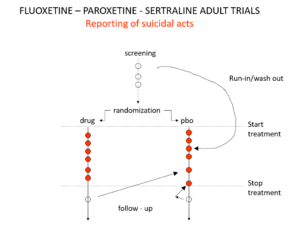
You’ve seen the son of this slide before. In all Antidepressant RCTs there was a 2-week washout period during which patients were whipped off prior medicines. We now know this was a tricky thing to do – it gives lots of suicides. But companies argued as the patients were on nothing, these events should be counted as placebo events – as the diagram here illustrates – along with events after the trial was over.
Slide 26
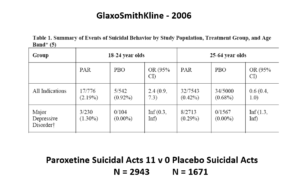
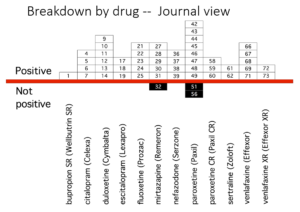
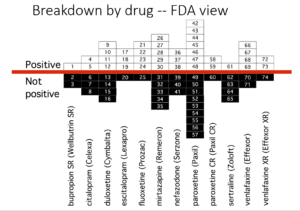
This slide shows data straight from a 2006 GlaxoSmithKline paper. GSK’s SSRI paroxetine was in trouble – the RCTs data for Major Depressive Disorder seem to show paroxetine causes suicidal events. The real data I think are worse that GSK admit to here.
Slide 27
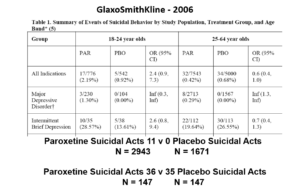
But never fear RCTs come to the rescue. GSK also did trials in people with Intermittent Brief Depressive Disorders – IBDD. These are borderline personality disorder to most people – patients who have suicidal events much more often than anyone else. But these patients can meet criteria for depression and could be entered into Depression RCTs.
Now Lilly had done a trial of Prozac in these patients – it didn’t work. GSK also did a trial of paroxetine which didn’t work and had a 3-fold higher suicidal act rate than placebo. GSK then did another trial in a similar group of patients. Why?
The answer is here. Here are IBDD data from the two GSK trials. I have seen other data for these two trials which make paroxetine look worse but let’s stick with GSK’s story. We could even add 16 more events to the paroxetine arm and still get the same magical outcome
Slide 28
When you add the IBDD data to the MDD data – all of a sudden paroxetine doesn’t cause suicidal events it protects against them. This is called Simpson’s paradox.
Something like this is going to happen in every trial of a treatment in a heterogenous condition – back pain, breast cancer, diabetes, hypertension, osteoporosis, parkinson’s disease. We can use an effect a drug causes to hide an effect a drug causes.
RCTs are not a good way to work out what is going on. It also means that statistical significance and confidence intervals have no anchor in the real world in a Clinical Trial.
Slide 29
In 1999 I was asked to speak at a company symposium in London – speakers would need to produce articles for a supplement. I said yes and soon after had an email with my article. It was a great Healy article saying the things Healy says in the way he says them with Healy references. No one who knew my stuff would pick it out as not mine.
I emailed back saying I figured on writing my own. There was surprise at the other end but they said okay. I sent it to them and they said this is rather good but there are some important commercial messages in the other one – we’ll get Siegfried Kasper to put his name on it.
Here it is – only one word changed from the original paper – the name of author Kasper. Everybody in Vienna knows this but it’s done no harm to SK’s career. You can find materials saying you can trust doctors like Kasper because they have written a 1000 articles or more.
Slide 30
A year later I was in Pfizer’s archive where even the loo-paper was stamped confidential. I came across this document covering the articles on Pfizer’s SSRI Zoloft being managed by Current Medical Directions – a medical writing company.
Slide 31
Inside there are pages listing the articles published or being written on Zoloft for the anxious, the depressed, the young, the old etc. Here’s the Post Traumatic Stress Disorder – PTSD – page.
On the right you see that the articles were already written for these essentially negative studies saying the drug worked wonderfully well. One would go to NEJM – the other to JAMA. And on the left – you see TBD – authors names are To Be Determined. Pfizer’s marketing department will work out who would be the best sales academics for the drug.
This is not just a mental health issue. It holds for all treatments across medicine.
Slide 32

Here is Study 329, a study of paroxetine v placebo in adolescent depression. It’s authorship line is to die for and it’s in the journal with the highest impact factor in child psychiatry and says paroxetine works wonderfully well and is entirely safe for children who are depressed. Any doctor reading this would race out to put all teens s/he could find and put them on this drug.
Slide 33

This internal GSK document from 1998 shows the company knew the trial had shown the drug didn’t work and proposed taking out the good bits of the data and publishing those which is the article you have seen. New York State took a fraud action against GSK on this basis. GSK were also fined $3 billion by the Dept of Justice, which led to access to the trial data and what you are going to see next.
Slide 34 The full story is on Study329.org
Slide 35
This legal action gave us access to company data no one ever sees. The efficacy data is pretty irrelevant, but it was still possible to show that no matter which way you cut the data paroxetine was not more efficacious than placebo.
Slide 36
The tricks used to hide the problems were the real interest in these data. The original article had 10 pages. Regulators see an 800-page Clinical Study Report (CSR) plus nearly 5000 pages of appendices – which no-one in FDA looks at. They now say they get Individual Patient Level Data – IPD – from companies. This is a spreadsheet with datapoints on individuals but these do not let regulators work out what is going on.
Behind these IPD in Study 329 we saw a further 77,000 Clinical Record Form (CRF) pages, and the material here was often inconsistent with the IPD. As you’ll see.
Point 2 points to data that didn’t get transcribed from the 77000 pages to the 5000 pages. Point 10 is patients on placebo got SSRIs – I can explain how. But I want to focus on coding issues.
Slide 37
The psychiatric adverse events all got grouped under neurological events – along with headaches and dizziness. This drowns out the signal from psychiatric adverse events.
We were sensitized to this by Elizabeth Loder, the BMJ editor handling our paper – which took over a year to publish with 7 review rounds and 7 reviewers – who objected to our every mention of headache. As it turned out she was a headache-ologist, who was an opinion leader for GSK but above all was the wife of an attorney working in Ropes and Gray who had been the lawyers defending GSK against the $3 billion fine.
Slide 38
If we strip out headache and dizziness, in the lower bar you see the number of suicidal events in the Keller paper – once you decode them from emotional lability. In the middle bar, GSK revised this after being asked to do so by FDA when a fuss blew up. In the upper bar you see that we found more again – and there were more than we missed as I’m about to tell you.
Slide 39
This slide points to something profound that has changed for all of us. It used to be accepted that people were moody and often went half crazy in their teens as they worked out what sense if any the world made. We thought it was important to go through this, as Homer tells Bart.
Now you get put on pills. There are 45 RCTs of antidepressants done for teens who are depressed – all negative. Even the two trials that led to Prozac being approved for children were negative. The 30 trials since 1990 have likely all been ghostwritten. They show the greatest divide in all of medicine between what the data shows and what the academic literature claims. RCTs are supposed to stop therapeutic bandwagons in their tracks but it looks like Antidepressants are now the second most commonly taken drugs by teenage girls and both these and stimulants are likely relatively commonly taken in this group.
And while Greta Thunberg and her generation try to clean up the environment, they are pumping more chemicals into their body than any prior generation in human history.
Slide 40
Here is a very famous photo. In a Pfizer trial of a psychotropic drug, a man poured petrol over himself and set a match to it, intending to kill himself. He died 5 days later from his burns. His death was coded as burns. But the company had to write a Serious Adverse Events narrative and if you saw that you could work out that he should have been coded as suicide.
Slide 41
Companies have found a way to get around the need to write a Narrative. Here is a young man on a street waving a gun. Its Kyle Rittenhouse. In Study 329 a boy of 15 was picked up out on the street waving a gun around and threatening to kill people. He was hospitalized and should therefore have had an SAE narrative but the company coded him as intercurrent illness.
Four children dropped out of Study 329 coded as intercurrent illness – all were taking paroxetine. Add them into the picture you have just seen and things look a lot worse.
What is intercurrent illness? This was almost certainly an adverse event on paroxetine but invoking an intercurrent illness that means you really should not have been entered into this study means there is no need to write a narrative. This loophole has been there for 25 years, and FDA have not moved to close it. It applies to all drug and vaccine trials also.
Slide 42
I told you the Prozac trials were negative. Here is part of FDA’s 2002 approval letter for Paxil. The key bit is typed up so you can read it. The date is important – the Keller paper was 2001.
You can see here GSK told FDA that Study 329 was negative and you see that FDA agreed to approve the drug on the back of three negative studies and also agreed that there was no need to mention this in the labelling. Why would FDA do this?
Slide 43
Here are the published results of adult trials of antidepressants nearly a decade earlier. The picture looks pretty good.
Slide 44
But as Erick Turner has shown, here is how these studies looked to FDA. A different picture. Companies don’t leave negative studies unpublished, they know FDA are happy to let them publish negative studies as positive. Why?
Perhaps GSK told FDA that if FDA let the world know Study 329 was negative, GSK might get sued for fraud – which they did and fined for $3 billion. FDA don’t feel inclined to blow this whistle.
Slide 45
In 1991, at the Prozac hearings FDA were sitting on data for sertraline and paroxetine showing the same limited efficacy compared with older antidepressants, and increased suicidal event rates on all three SSRIs with all companies breaching FDA regulations to hide the problems.
FDA opted not to warn because warning would put people off seeking the benefit – from a treatment that according to them had a favourable benefit-risk ratio even though more people were dying on the active treatment than on placebo, and the adult benefits were not clearly different to the benefits in children.
From this point on the hazards of drugs began to vanish. Journals like the BMJ became scared to take case reports which previously had been the bedrock of medicine. Drugs and Therapeutics Bulletins vanished – replaced by Guidelines which only listed benefits.
Doctors have now lost a sense for drug induced toxicity, which is often coloured by anxiety or depressive symptoms. Faced with someone saying they feel anxious or depressed as you can do when you have the Flu, are pregnant, or have a toxic reaction to a drug, doctors increasingly give more of the drugs that are causing the problems in the case of psychotropic meds.
We have become Invisible to our doctors, not heard or seen the way we once were.
If we become invisible, healthcare staff do too. Managers don’t see or hear them dealing with toxicities that need expert input. Managers see drugs the evidence says work well and are free of problems and figure doctors can be replaced with cheaper prescribers – or perhaps robots soon. And, if things go wrong, it must have been the doctor’s fault.
When you are faced with a patient who thinks they have become suicidal on treatment – the first scientific task is to work out what is happening. This is a judicial task. Key to this is the conversation with the patient who may say ‘Doc I’ve been depressed and suicidal before but this was different – I think it’s the drug – it cleared when I lowered the dose’.
This is a scientific engagement with the data – the person. The one time you have access to all the data is at this point. If you conclude the drug has made the person suicidal but the published academic adverts do not support this, the next scientific task is to reconcile the mismatch.
In the case of SSRIs and suicide, you find:
- companies breached FDA regulations in hiding events
- companies misapplied statistical methods
- the entire literature on these drugs is ghostwritten
- there is no access to the people in these trials for independent scrutiny
- FDA don’t have and have never seen the data
Slide 46
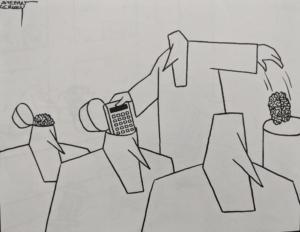
If you Follow the so-called Science you end up in brain transplant country. As this cartoon from over 40 years ago shows, this is Mindlessness. When we get to the point of A.I. we will have arrived at a moment of real peril.
The Art of Medicine is not the soft bed-side manner side of Medicine with RCTs being the Science. The Science of Medicine lies in making hard judgement calls. The Artefact of medicine makes your life easy but will doom you and us. There cannot be an us if you follow the artefact.
There are 3 ways out.
Slide 47
Donald Trump weaponized the phrase Fake literature – the greatest concentration of Fake Literature now centres on the drugs your doctor gives you.
He also gave me another good idea. After a school shooting, he said we need a good guy with a gun outside all these schools. People were horrified but had no easy answer to the follow up – well if a good guy with a gun isn’t a good idea why do we have them outside the White House?
Drugs are like guns – they can be very helpful in certain situations. But we sense that letting them multiply up and leak into every situation is a recipe for problems.
Drugs and Guns are techniques – gadgets. All gadgets create an arms race. Whether the use of these gadgets enhance or diminish us is down to us – but if we stop thinking about what we are doing when we use them we are highly likely to be diminished.
Because of the gadgets we call RCTs, the drugs race is not about creating more effective chemicals – it’s about creating more effective propaganda. By definition this diminishes us.
Slide 48
The arms race brings out an important message. Efficacy has its limits. We can make weapons more and more effective to the point that they can’t be used.
The same holds true for drugs. If you are on more than 3 medicines, effectiveness diminishes. To get the most effectiveness you need to be on 3 or less. Over 40% of over 45s are on 3 or more drugs every day of the week – this figure includes the people who never come to see doctors and over 40% of over 65s are on 5 or more drugs every day of the week.
We know that life expectancy even before Covid was falling. We know that reducing medication burdens can increase life expectancy, reduce hospitalizations and improve quality of life.
Paring our rapidly escalating medication burdens back to 3 or less should mean taking the values of the patient into account – but we are not doing it.
We introduced RCTs hoping to use efficacy to improve safety. We’ve done just the opposite. Now there is a chance to improve safety by deprescribing in order to get efficacy.
Slide 49
Deprescribing tries to smuggle safety in the back door. If you knock on the front door and ask a doctor to help you work out what is happening on a drug, the climate in which you both will have to do this now claims hysterically that you are being subjective, misinformed and crazy if you don’t Follow the Science.
The AstroScience we now have has converted both you and your doctors into thinking drugs are sacraments – things that can only do good. Up to 1990 we thought drugs were poisons and the magic of medicine lay in bringing good out of the use of a poison but to do that I had to know I was giving you a poison and you had to know you were taking one.
The magic used to lie in you and me doing science – now it lies in the pill or the vaccine. Me and you doing Case Based Medicine offers the only science in clinical psychopharmacology.
One of my patients years ago dropped out of High School early, and had no background in healthcare, when she was put on an SSRI. She ended up convicted of drunk and disorderly behaviour, crashed her car, was jailed, lost her job. She figured it was the SSRI she had been put on. Her doctor and AA told her this line of thinking proved she was an alcoholic.
She was right and they were wrong. It took years but she went on the internet and ended up being able to tell me things about the serotonin system and what drug she should be put on instead of an SSRI. Few people knew what she had found – other than the pharmaceutical industry who were busy working on just this as possible treatments for alcoholism.
Rather than seeing 100 heart-sink patients in my waiting room, heart-sink because they don’t do what I tell them to do – I could see 100 free researchers who could help me discover things I never knew about. The job could be fun.
What I got from this is that Motivation is worth more than Expertise.
Slide 50
Here is Walter Raleigh convicted on the basis of Hearsay. If your doctor reports your problem on a drug to FDA or CDC, FDA remove identifying names. This converts the report into Hearsay – useless to you or anyone who has been similarly injured by the drug. Unless you can be examined and cross examined there is no way to establish what has happened. There is a lot of talk today about misinformation and people ask where it comes from – well FDA and CDC create it.
Slide 51
If your doctor even listens and reports . Chances are he will get nasty if you bring a problem – tell you, this is all in your mind. In the case of a problem like PSSD, he will say a drug that is out of your body for months cannot cause this.
Any doctor who is nasty is so ultimately because he is scared. If he admits a drug can cause a problem, he fears the regulators and guideline makers, and his colleagues and politicians and the media will be nasty to him. Even Greta Thunberg’s generation will mobilize against him. So, instead of him, you will feel the violence built into the system.
Patients with PSSD often go on to commit suicide. It’s partly because of the horrific problem. It’s partly also because of the ridicule they get at from doctors. And a deep sense that they are not just the victims of a drug but victims of a miscarriage of justice and science.
Slide 52

So this lady is in my waiting room. She is smelly and hunched and grim to look at. She has come to me with a problem. She has been on an SSRI and she now cannot make love. I tell her sure temporarily while on the pills you might not be able to make love but if you want to go away for a romantic weekend you can stop.
She says I have been off my SSRI for 3 months and I can take a hard bristled brush and rub it up and down my genitals and feel nothing. Here is a scientific riddle the answer to which is worthy of a Nobel Prize. Its not just finding out something new about biology – the answer will likely change our entire view of ourselves.
This lady in fact comes from an Arthurian Legend – Gawain and the Loathly Lady. Arthur has been trapped by a Black Knight who spares his life if he can answer a riddle – What do Women Most Desire. Having hunted the answer for months, Arthur and his court despair of finding the answer. Two days before he is due to die, Arthur and his troop meet this woman by the edge of the road who tells him that she can give him the answer to the riddle but one of his knights must become her husband. Gawain jumps down and offers himself up.
Slide 53
Gawain gets married. Everyone at the Court is desperately unhappy for him.
Slide 54

When they go up to the bedchamber Gawain can’t even bear to look at her. He turns and finds the most beautiful lady. She asks him – do you want me to look like this in the bedchamber or during the day in court. He has no idea what to say and says – whatever you want. Which turns out to be the right answer.
She like us wants to control her own life. Give her or you this and s/he or you can become the most interesting and, in that sense, attractive person imaginable. She wants your support to live the life she wants to live. There may be a disease that needs treating – but she doesn’t want you to tell her how to live a life, or want her negative emotions eliminated with a pill. She may be doing better at living life than you or I.
I’ve talked about deprescribing – a smart solution to our problems. I’ve talked about putting our name to adverse events – a solution that involves courage.
But here is another very strange and hard to put in words element – you have to believe in people. Science depends on collaboration with others. The more you collaborate the more interesting and the more you like the people you collaborate with, which leads them to like you too because you like them.
Science and democracy and love are intimately entwined.
Science does not thrive in the presence of absolute power, absolute truth, or where money dictates.


































P
nice article
I have colleagues that were very upset when bombed on Zoom
Dear Dr Healy,
Two points, the shennanegans of the pharmaceutical industry do not only apply to SSRI’S but the whole spectrum of psychiatric chemicals. As well as ECT.
Please be more forthright.
And thank you.
Seasoned DH Followers will love this blog.
Personally, I would love to hear the Q & A to establish how much of this content they took in and understood and to give us a measure of their reactions to it.
This blog tells the story and gives an indication of why we are not believed – Even GSK $3B fine didn’t cut the mustard and didn’t see the earth quake…
The Perfect Killing Machine
https://www.youtube.com/watch?v=2RnPN0pAbX8
Zoom-bombing upsets Queens University
According to a group of Canadian researchers, “Zoom bombing” became popular when the pandemic began.
https://globalnews.ca/news/7621278/queens-university-lecture-zoom-bombers/?utm_source=headtopics&utm_medium=news&utm_campaign=2021-02-05
Why Zoom-bomb Who’s Afraid of Science?
A later post will say more. …
“Standardization without judgement calls gives us Big Mac hamburgers rather than really good hamburgers.”
When I read these words immediately I thought about the state of undegraduate education in America, which is becoming increasingly McDonaldized. The prevailing ideology seems to be this is a job any dolt can do about as well as any other, provided he or she is sufficiently micromanaged and controlled by technology.
FTR, before anyone starts kicking over straw men, I am not saying the people who work for McDonald’s are dolts — I am just saying this is how they are treated by their overlords. And I have nothing against McDonald’s — I just don’t think their business model is a good one for what I thought we were here to do, which is to turn out more competent and capable citizens.
I have always found consolation in the knowledge that I am getting paid to do what I love. But it seems that every year I spend less and less time doing that, and more and more time ticking boxes and jumping through hoops invented by people who get paid (a lot more than I do) to come up with boxes for me to tick and hoops for me to jump through.
I logged on at 11pm UK time. It is/was a great presentation. I felt for Professor Healy and YM when an absolute ‘Twat’ sort of gate crashed the event, hell bent on upsetting and maybe shocking the viewers. I would not give him the satisfaction, and am glad to see the rest of the presentation here. Mining this talk, reveals gold nuggets everywhere. All medics, Scientists, ethicists should delve into these, invaluable, at times brutal insights. I love the Loathly Lady story… In a post #Me too era.. How vital it is that all women (and Men) are believed when they tell uncomfortable truths and that men accept that by supporting those truths life will be better for all. Thank you Professor Healy for your relentless research and enduring courage. You are an inspiration to many. Like Annie, I too would be keen to see a Q & A session.
Thank you for a moving and eloquent plea for sanity.
To gatecrasher – I would honestly like to view the ‘gatecrashing’ to see what was bugging you to such an extent, There is limited time to discuss in Q and A’s so may I ask if you would be prepared to help those of us who support DH’s scientific work , his humanity, attitude of kindness and democracy, understand your opposition? A variety of views are published on the blogs – commentators are not simply ‘groupies’ but interested people with concerns in common . New perspectives are possible in any area of life but need to be offered respectfully even if angrily if they are to be listened to respectfully in an open minded way.
Maybe the gatecrasher was just a paid pharma troll.
“It is theft on the part of GoFundMe,” Mr. Cruz said. “Let me say, the Canadian truckers are heroes. They are patriots marching for your freedom and for my freedom.”
https://www.washingtontimes.com/news/2022/feb/7/ted-cruz-demands-federal-probe-gofundmes-suspensio/
Ottawa declares State of Emergency
https://www.youtube.com/watch?v=Jp8slGg7O7M
The Honkening
https://www.youtube.com/watch?v=SNKcrzopee0
It’s time to end the ‘freedom convoy’ in Ottawa by enforcing the law and following the money
MARK CARNEY
https://www.theglobeandmail.com/opinion/article-mark-carney-end-freedom-convoy-ottawa-state-of-emergency/
But now in its second week, no one should have any doubt. This is sedition.
Truly shocked at Mark Carney …
Thank you for sharing another gifted and inspiring lecture Professor Healy. I am intrigued to await the Q and A’s response. Also to learn more about the unwelcome interruption. We need your information to be received and understood by the widest possible global audience. In the UK, it is ‘Child Mental Health Week’. As usual, conventional, drug-based psychiatry is afforded extensive media propaganda opportunities, and is never subject to being challenged regarding its appalling outcomes, especially iatrogenic ‘suicide’. A recently formed UK Suicide Prevention Charity is also receiving much media publicity here. It is heart breaking to listen to those devastated by the loss of their children and their tormented bereavement. However, when I searched their website, there was nothing found in response to typing in: ‘AKATHISIA’ – ‘SSRIs’ or ‘Antidepressant Induced Suicide’. How can suicide prevention be comprehensively addressed by such sincere and committed campaigning if these concepts remain unknown or undisclosed?
Is that “Papyrus”?
‘Canadians trust science,’ Trudeau said
Justin Trudeau
@JustinTrudeau
·Officiel du gouvernement – Canada
Canadians have the right to protest, to disagree with their government, and to make their voices heard. We’ll always protect that right. But let’s be clear: They don’t have the right to blockade our economy, or our democracy, or our fellow citizens’ daily lives. It has to stop.
Justin Trudeau
@JustinTrudeau
Officiel du gouvernement – Canada
So far, hundreds of RCMP officers have been mobilized to support the Ottawa Police Services. We’re also working with municipal partners to further strengthen our response, and we’ll continue to be there with whatever resources are needed to get the situation under control.
https://twitter.com/justintrudeau
Trudeau stokes divisions by branding the 7,000-strong Freedom Convoy in Ottawa as ‘swastikas wavers’: Anti-mandate protesters blockade busiest border crossing to US at Detroit
https://www.dailymail.co.uk/news/article-10486929/Canada-pushes-against-GOP-support-COVID-protests.html
‘Canadians trust science,’ Trudeau said. ‘A few people shouting and waving swastikas does not define who Canadians are.’
Who’s Afraid…
“The Covid dashboards are a great mechanism to generate perceptions of risk and fear. The vaccines of course treat risks – not disease.”
I have posted an analysis of US 2021 deaths ages 15-64 on my blog and have concluded that _at least_ 137,500 and maybe twice that have died from the covid vaccines in the heart disease, cancer, and stroke categories.
An article quoting an insurance executive had the increase in deaths of working age people at 40% in 2021 over 2020. And he asserted that the increase had nothing to do with covid.
https://www.thecentersquare.com/indiana/indiana-life-insurance-ceo-says-deaths-are-up-40-among-people-ages-18-64/article_71473b12-6b1e-11ec-8641-5b2c06725e2c.html
That leaves covid vaccines by the process of elimination.
Thanks for this article, Pilot! A couple of things I noticed:
The 40% increase in deaths among working-age people cited by Mr. Davison isn’t an increase of 2021 over 2020 – it’s an increase of 40% over “pre-pandemic levels.” That includes the whole period since March 2020, both before the vaccine rollout and after it. Total deaths had already risen 23% in that pre-rollout period (March-December 2020).
I agree that vaccine-related deaths have likely been way under-counted. But they’re NOT the only explanation for increased deaths “other than Covid-19.” Far from it. The damage done by pandemic shutdowns, on top of the virus itself, has been huge. Deaths from diabetes, Alzheimer’s and heart disease all rose in those first ten months. Murder rates rose sharply; suicides were up as well. Overdose deaths, mostly opioids, hit a stunning 100,000 in 2020. Not to mention the deaths that always go with mass unemployment.
People concerned about mRNA vaccine harm need to understand these numbers. Why? People’s willingness to tolerate vaccine risks will depend on how dangerous we believe the virus is – both to our community, and to us personally. Not every precaution is worth the harm it may cause, whether it’s a vaccine or a school shutdown.
Covid-19 has killed a hell of a lot of people. But the current risk is less than in March 2020 and will vary by each person’s age, sex, health, etc. An un-vaccinated 21-year-old might be “higher risk” than his vaccinated buddies, but “lower risk” than his 60-year-old vaccinated uncle with diabetes. At the same time his risk of vaccine side effects is probably higher than his uncle’s.
My local newscast just announced that with a possible end to school mask mandates, “some parents are jumping for joy, while others are terrified.” Neither group of parents are stupid, or evil. But it’s like we live in separate realities, fed by separate streams of propaganda. We can’t go on like this. If we want our fellow citizens to look critically at vaccines, we need to demand a sober, honest accounting of the Pandemic as a whole.
“The 40% increase in deaths among working-age people cited by Mr. Davison isn’t an increase of 2021 over 2020 – it’s an increase of 40% over “pre-pandemic levels.””
Yes, that’s correct. I should have been clearer.
Just to be clear, the numbers show that murders, suicides, and overdoses for both 2020 and 2021 are within pre-pandemic historical ranges. Still, the panicdemic and the postponement/rescheduling of “elective” procedures likely contributed somewhat to deaths due to delayed health care.
Working age deaths were up somewhat in 2020 (+77,000), but rose _sharply_ in 2021 (+137,500)–and the 2021 numbers are still preliminary and are likely to increase further as statistics are gathered.
There was a Nature article reviewed by Clare Craig where Craig showed that covid deaths in older men from myocarditis were around 1 per 3,000 cases and vaccine deaths in older men from myocarditis were around 1 per 5,000 vaccinations. So I would expect a significant contribution to excess mortality in working age people from chronic complications of covid as well as from vaccines.
Looking at 2021 US working age excess mortality causes of death is a complicated business. But two things stick out–in 2021, deaths in the 85+ y.o. group were down 14% from 2020. And deaths from chronic lower respiratory disease were down 12% from 2020. We would expect deaths from other geriatric causes (heart disease, cancer, stroke) to be down similar to the decline in chronic respiratory disease if deaths in other age groups held steady. But deaths from other geriatric causes didn’t decline and the only explanation is that geriatric deaths in working age people must have increased.
There are two explanations that I see likely–that some of the deaths were contributed by complications from covid and some by vaccines. Maybe also a small percentage by people missing some health care.
Hot off the presses from the Department of Homeland Security:
Questioning the pharmaceutical companies’ narrative is a “key factor” contributing to the “current heightened threat environment.”
Holy shit.
https://www.dhs.gov/ntas/advisory/national-terrorism-advisory-system-bulletin-february-07-2022?fbclid=IwAR338zJFTX2Ky44nMQAYLVnRSdDMRhRg5dEJFhQmQ_aYhyT4JcuU_xfRRP0
Thanks Patrick.
Department of Homeland Security:
‘ACTIVE SHOOTER AND HOW TO RESPOND’ – RECOGNISING POTENTIAL WORKPLACE VIOLENCE.
Missed opportunity? No reference: HOW TO RECOGNISE AKATHISIA, which together with disinhibition and emotional blunting MUST be considered as a potential, causative factor in VIOLENCE against self and others.
Biden urges Trudeau get even TOUGHER with Freedom Convoy: Ontario authorizes cops to seize vehicles, fine drivers and asks court to freeze $8.6M donated on GiveSendGo as mayor warns truckers will be ‘physically removed’
https://www.dailymail.co.uk/news/article-10501225/Just-three-days-honking-ban-Ontario-brings-draconian-rules-Freedom-Convoy-truckers.html
Canadian Freedom Truckers Protests Spread Across The World
https://www.youtube.com/watch
https://www.youtube.com/watch?v=yYVFRftAUeI
Testimony of Dr. Joan-Ramon Laporte Roselló to the Spanish Congress’ Vaccine Commission
http://coronacases.wiki/index.php/wiki/pdf-testimony-drrosello/
Dr. Roselló references Rxisk
SPAIN: Dr. Roselló in Congress: so-called mRNA “Vaccines” Are NOT Vaccines
https://www.ukcolumn.org/community/forums/topic/spain-dr-rosello-in-congress-so-called-mrna-vaccines-are-not-vaccines/
Justin Trudeau@JustinTrudeau
Officiel du gouvernement – Canada
I convened the Incident Response Group again today – we went over the efforts underway to address the illegal blockades and occupations. We’ll continue to make sure municipal, provincial, and federal authorities have what they need to end the blockades and protect public safety.
The Prime Minister stressed that border crossings cannot, and will not, remain closed, and that all options remain on the table.
https://pm.gc.ca/en/news/readouts/2022/02/12/prime-minister-justin-trudeau-convenes-incident-response-group-ongoing
https://www.youtube.com/watch?v=pXsaePtXqTE
Who’s Afraid – of GSK’s involvement in the manufacture of this vaccine…..
for some history of GSK. read exposure by Doshi and colleagues in 2013 published on DH blog 2015
https://davidhealy.org/a-milestone-in-the-battle-for-truth-in-drug-safety/
and on wikipidia …UK found ‘insufficient evidence’ in 2019
GSK China scandal
From Wikipedia, the free encyclopedia
The GSK China scandal was an imbroglio where the China branch of the global drugmaker GlaxoSmithKline (GSK) pleaded guilty to paying bribes to doctors and hospitals to promote the company’s products in China.
The scandal started with sex tapes of the company’s China head, Mark Reilly and his Chinese girlfriend at a Shanghai department, which were sent to several senior executives of the company. The company’s investigations into the sex tapes led to imprisonment of the paid investigator Peter Humphrey and his wife Yu Yingzeng in China, a country which has an opaque judicial system with a 99% conviction rate, on allegations that they had breached privacy law. Humphrey and his wife maintain their innocence and Humphrey claims he was tortured and denied medical treatment due to his refusal to confess to trumped up charges.
After the trial in Changsha in September 2014, the company apologized to the Chinese people, and paid one of the biggest fines in Chinese history worth ¥3bn (£300m; €350m; $490m). 4 executives of the company, including Mark Reilly, the only foreign citizens involved, were sentenced. Reilly received a suspended sentence and was also deported from China. The UK Serious Fraud Office concluded their investigation in 2019 after finding insufficient evidence[1]one of many publications about massive scandals
and
https://www.drugwatch.com/manufacturers/glaxosmithkline/Drugwatch.co
Last Modified: March 24, 2021
Like many pharmaceutical companies, GSK has stepped up to develop treatments for COVID-19. In March 2021,
…..But GSK has faced allegations of unethical and unsafe business practices. Some of its former employees were criminally prosecuted on two different continents.
Some of GSK’s popular pharmaceuticals have led to product liability lawsuits. These include Avandia, Zantac, Paxil, Prevacid 24HR, Nexium 24HR and Zofran.
Medicago and GSK
phot A worker inspects the Nicotiana benthamiana plants at Medicago greenhouse in Quebec City, August 13, 2014.
Update
Dec 7 (Reuters) – Canadian drug developer Medicago’s plant-based COVID-19 vaccine candidate, enhanced by GlaxoSmithKline’s (GSK.L) booster, was 75.3% effective against the Delta variant of the virus in a late stage study, the two companies said on Tuesday.
They said the vaccine’s overall efficacy rate against all variants of the coronavirus was 71%, except Omicron, which was not in circulation when the study was underway.
Last week, infectious disease expert Anthony Fauci said though the focus is on Omicron, 99.9% cases in the United States were due to the Delta variant. “Delta continues to drive cases across the country, especially in those who are unvaccinated.”
Medicago will imminently seek regulatory approval for the world’s first plant-based COVID-19 vaccine from Health Canada as part of its rolling submission.
The Canadian health regulator in April accepted Medicago’s application for a real-time review of the only home-grown COVID-19 vaccine to reach the most advanced level of trials.
Medicago, which uses GSK’s adjuvant to boost the efficacy of its vaccine, started the late-stage study in March with over 24,000 participants aged 18 years or above in Canada, United States, United Kingdom, Mexico, Argentina, and Brazil.
Trials in Wales (United Kingdom)
Swansea volunteers first in Europe to come forward for Covid vaccine trial
May 17, 2021
Wales to take part in UK trial for plant-derived Covid-19 vaccine
The vaccine has already been through early phase human studies and now requires testing on a large scale and a study involving 1,500 people across the UK is starting.
CoVLPs do not have any viral genetic material and cannot therefore cause the disease. The CoVLPs are combined with an adjuvant before the vaccine is given. An adjuvant is an ingredient that may enhance the body’s immune response to the vaccine and allow for a smaller dose of the vaccine to be given so that more doses would be available to vaccinate more people once the vaccine is approved.”
The Medicago vaccine study will run across 14 sites in the UK in addition to multiple sites in the United States, Canada, Europe, and Latin America. The study will enrol up to 30,000 volunteers worldwide.
“Medicago’s plant-derived Covid-19 vaccine has already been through early phase human studies.
07 December 2021 (Extracts)
Medicago and GSK announce positive Phase 3 efficacy and safety results for adjuvanted plant-based COVID-19 vaccine candidate
For media and investors only
Issued: London UK and Quebec City, Canada
Primary endpoints and secondary endpoints for which data are available were met in trial dominated by COVID-19 variants
Efficacy demonstrated against all variants seen in the study, including 75.3% efficacy against COVID-19 of any severity caused by the globally dominant Delta variant
Vaccine candidate was well-tolerated, with no related serious adverse events reported in the vaccine group
Final regulatory submission to be filed with Health Canada imminently
Medicago, a biopharmaceutical company headquartered in Quebec City, and GlaxoSmithKline plc (GSK) today announce positive efficacy and safety results from the global Phase 3 placebo-controlled efficacy study of Medicago’s plant-based COVID-19 vaccine candidate in combination with GSK’s pandemic adjuvant, conducted in over 24,000 subjects (adults 18 years and above) across six countries.
…
During the study, no related serious adverse events were reported and reactogenicity was generally mild to moderate and transient; symptoms lasting on average only one to three days. To date, the Phase 3 results have confirmed the safety profile is consistent with Phase 2 results. The frequency of mild fever was low (<10%), even after the second dose. Full results of the Phase 3 study will be released in a peer-reviewed publication as soon as possible.
Based on these results, Medicago will imminently seek regulatory approval from Health Canada as part of its rolling submission. The vaccine candidate is not yet approved by any regulatory authority.
“This is an incredible moment for Medicago and for novel vaccine platforms. The results of our clinical trials show the power of plant-based vaccine manufacturing technology. If approved, we will be contributing to the world’s fight against the COVID-19 pandemic with the world’s first plant-based vaccine for use in humans,” said Takashi Nagao, CEO and President at Medicago. “I want to thank everyone who participated in our clinical trials, our collaborators at clinical trial sites, our partners at GSK, the Government of Canada and Government of Quebec, all of our employees and Mitsubishi Tanabe Pharma Corporation, for their commitment to advancing vaccine science when the world needs it.”
Medicago has been developing its plant-based technology for the past 20 years, using unique technology to produce Virus-Like Particles (VLP) for its protein vaccines. VLPs are designed to mimic the native structure of viruses, allowing them to be easily recognised by the immune system. Because the VLPs lack core genetic material, they are non-infectious and unable to replicate. VLP vaccines developed by other technologies have traditionally been used worldwide for more than 30 years.
Medicago has initiated the regulatory filing process for the adjuvanted plant-based COVID-19 vaccine candidate with the FDA (US) and the MHRA (UK). Preliminary discussion is underway with the WHO for preparation of the submission. Medicago has also initiated a Phase 1/2 trial in Japan where it plans to submit for regulatory approval in combination with the Phase 2/3 global study results next spring.
About the Phase 2/3 study
The Phase 2/3 study has a multi-portion design to confirm that the chosen formulation and dosing regimen of the vaccine candidate (two doses of 3.75 µg of antigen combined with GSK's pandemic adjuvant given 21 days apart) has an acceptable vaccine profile in healthy adults 18-64 years of age, elderly subjects aged 65 and over and adults with comorbidities.
. All subjects are being followed for a period of 12 months after the last vaccination for assessment of safety and the durability of the immune responses which will be the final analysis.
The Phase 3 portion of the trial was launched in March 2021 and was an event-driven, randomized, observer-blinded, crossover placebo-controlled design that evaluated the efficacy and safety of the vaccine candidate formulation, compared to placebo, in over 24,000 subjects across Canada, the United States, United Kingdom, Mexico, Argentina, and Brazil.
About Medicago
Medicago is on a mission to improve global public health using the power of plants. Founded in 1999 with the belief that innovative approaches and rigorous research would bring new solutions in healthcare, Medicago is a pioneer in plant-derived therapeutics. We are proudly rooted in Quebec, with manufacturing capacity in both Canada and the US.
Medicago is an affiliated company of Mitsubishi Tanabe Pharma Corporation.
07 December 2021
Medicago and GSK announce positive Phase 3 efficacy and safety results for adjuvanted plant-based COVID-19 vaccine candidate
For media and investors only
Issued: London UK and Quebec City, Canada
75.3% efficacy against COVID-19 of any severity caused by the globally dominant Delta variant
Vaccine candidate was well-tolerated, with no related serious adverse events reported in the vaccine group
Final regulatory submission to be filed with Health Canada imminently
Medicago, a biopharmaceutical company headquartered in Quebec City, and GlaxoSmithKline plc (GSK) today announce positive efficacy and safety results from the global Phase 3 placebo-controlled efficacy study of Medicago’s plant-based COVID-19 vaccine candidate in combination with GSK’s pandemic adjuvant, conducted in over 24,000 subjects (adults 18 years and above) across six countries.
Full results of the Phase 3 study will be released in a peer-reviewed publication as soon as possible.
Based on these results, Medicago will imminently seek regulatory approval from Health Canada as part of its rolling submission. The vaccine candidate is not yet approved by any regulatory authority.
Medicago has also initiated a Phase 1/2 trial in Japan where it plans to submit for regulatory approval in combination with the Phase 2/3 global study results next spring.
The Phase 3 portion of the trial was launched in March 2021
GSK is collaborating with several organisations on COVID-19 vaccines by providing access to our adjuvant technology. We are working with Sanofi, Medicago and SK bioscience to develop adjuvanted, protein-based vaccines, and all are now in Phase III clinical trials.
About Medicago
Medicago is a pioneer in plant-derived therapeutics. We are proudly rooted in Quebec, with manufacturing capacity in both Canada and the US.
Medicago is an affiliated company of Mitsubishi Tanabe Pharma Corporation.
.
This was a most astonishing presentation. It would be great if a paper translation of the longer version of the video could be made widely available to all in all countries . (The google translation of the video is very poor, it’s difficult to read ie ‘Longer video version Length: 29:54
Turn on closed captions “CC” and “Settings”>subtitles>auto-translate>English’
With huge gratitude to Dr J-R Laporte Rosello who also produced a document published by D H on Rxisk blog as early as 2 years ago
Medications compromising Covid Infections
April 2, 2020 | 74 Comments
IN THE MIDST OF THE SARS-CoV-2 PANDEMIA, CAUTION IS NEEDED WITH COMMONLY USED DRUGS THAT INCREASE THE RISK OF PNEUMONIA.
Joan-Ramon Laporte, M.D.
Emeritus Professor of Clinical Pharmacology, Department of Pharmacology, Therapeutics and Toxicology
Universitat Autònoma de Barcelona.
Fundació Institut Català de Farmacologia. WHO Collaborating Centre for Research and Training in Pharmacoepidemiology.
jrl@icf.uab.cat
David Healy MD FRCPsych
Professor Dept of Family Medicine
McMaster University
Joan-Ramon took the initiative in creating this document and pushing for its rapid publication. It will also be published on No Gracias and other platforms and we will seek other translations. A PDF version of the article in English can be downloaded from HERE and in Spanish HERE.
“NHS KILLED MY MOTHER WITH MIDAZOLAM”
https://www.bitchute.com/video/8nt3gYWvExk8/
Laporte in Court, arguing scientifically about covid19 vaccines.
Gervas, John
https://www.actasanitaria.com/opinion/el-mirador/laporte-en-corte-argumentando-cientificamente-acerca-vacunas-covid19_2001063_102.html
The intervention of Joan R. Laporte, an expert in pharmacology, in the Congress of Deputies on the vaccination of covid19 caused a real commotion whose foundations our collaborator analyzes.
Freedom of expression
Profound changes for humanity have been brought about by being able to nourish itself regularly, safe access to safe drinking water (and its purification), formal education from childhood and the social structure that facilitates a certain equality, also supporting the necessary freedom of scientific, political and religious expression.
In pericles’ classic Athens, freedom of expression was achieved with
1/ isegoria, which gave equal right to speak to every citizen, and to be considered for the basis of their contributions, not for their value in the expression of the same and
2/ parrhesía that gave the right even to defend ideas boldly, even if they seemed extravagant, and against the powerful, to the point of ridiculing them.
That is, it ensured at the same time the possibility of speaking and doing so frankly.
In the response to the covid19 pandemic we have failed with respect to such freedom of expression, tramutated for the sake of a barbaric “savior” health that entails a unique thought.
There has been and is not isegoria; nor, parrhesía. Frankness has been ridiculed by transforming their proposals into “hoaxes” (fake news), no matter how much they are published /published in the best scientific journals in the world, and those who disagree/disagree are labeled with the stigma of mental illness, assigning them various phobias, from denialists to anti-vaccines to flat earthers, even if they were even professors at prestigious universities.
In the response to the covid19 pandemic we have failed with respect to such freedom of expression, tramutated for the sake of a barbaric “savior” health that entails a unique thought.
This unique thought is always justified with the opinions of experts, in whom the basis is found to justify the unjustifiable, such as the use of the mandatory muzzle outdoors and the promotion of covid19 vaccines as life-saving. With experts, politicians justify their decisions and impositions, and Medicine becomes religion and magical activity, and its proposals, talismans.
That’s why people are still wearing the muzzle on the street, because masks have been transformed into talismans.
With the same fervor, covid19 vaccines and re-vaccines are accepted and required, as life-saving fetishes, which will protect against evil, even irrationally.
The Fallacy of Death Avoided by Medicine
There are those who continue to believe scientifically that Medicine and its advances and medicines have managed to change the face of the Earth allowing the progress of Humanity.
It is a belief like any other since Medicine provides 10% of the improvements, at most, and only with respect to health. With this belief, magical narratives are created. For example, it is claimed that scientifically speaking, vaccines and doctors save lives.
It is a fallacy that of “death avoided”, because no one can prevent humans from ending up dead.
Vaccines (and doctors), if anything, change causes of death and prolong lives. It’s no small thing, no.
The arrogance of experts that muzzles freedom of expression
We have lost freedoms producing immeasurable damage, including avoidable deaths.
Who will take to court for conviction and reparation of damages to those guilty of action and omission (experts and politicians, scientific societies, etc.) of confinements, school closures, curfews and other paraphernalia of barbaric health?
https://www.abc.es/sociedad/abci-confinamientos-restricciones-tenido-efecto-reduccion-mortalidad-covid-19-202202021455_noticia.html
It is the (pseudo) experts who have been blowing against logic, with the consequent dead.
David Sackett made it clear, when accurately criticizing preventive medicine, and therefore vaccines:
“Preventive medicine presents the three elements of arrogance:
1/ The first is that she is aggressively assertive, chasing people without symptoms and telling them what to do to stay healthy. Sometimes and relying on the value of the Law (vaccines, seat belts), it prohibits and prescribes patients and the general public of any age and condition.
2/ Second, preventive medicine is presumptuous, confident that the interventions it will adopt, on average, will do more good than harm to those who agree to follow them.
3/ Finally, preventive medicine is arrogant, lashing out at those who question the value of its recommendations” https://rafabravo.blog/2019/08/08/la-arrogancia-de-la-medicina-preventiva/
Laporte’s concerns about adverse effects and ineffectiveness of measures against the pandemic, including covid19 vaccines, are those of the best professionals and scientists in the world
And being in this pride of the single thought, Joan Ramon Laporte arrives at the Spanish Parliament, at the Pacata and Provincial Court in Madrid, and exercises the freedom of expression of classical Athens
Indeed, on Monday, February 7, 2022, professor and pharmacovigilance expert Joan Ramon Laporte, a Catalan figure of international prestige
http://www.nogracias.org/2022/02/09/transcripcion-y-video-de-la-intervencion-de-joan-ramon-laporte-en-el-congreso-de-los-diputados-relativa-a-la-gestion-de-las-vacunas-covid-y-el-plan-de-vacunacion-en-espana/
His freedom of expression led to a real political-scientific storm, and his consequent lynching.
Thus, they intend to transform him into an old man anchored in the old forms of pharmacovigilance, good in his time, but incoherent in the current one.
As Sergio Minué has highlighted, this lynching is not accidental or exceptional: “Laporte is not the only example. The same lynching has suffered or suffers people like John Ioannidis, Peter Gotzche, Alyson Pollock, Carl Henegan. Anyone who dares to question “true religion.”
And apostille: “It’s really scary post-pandemic winter.”
https://gerentedemediado.blogspot.com/2022/02/atticus-finch-y-la-jauria-mediatica.html
There are so many experts who have become a deadly plague
“Laporte’s intervention was respectful, clear and documented in each and every one of his statements”, very exact definition of the intervention, says Josep Casajuana.
And he continues: “Laporte often “provokes” because what he says does not conform to “normality” but his arguments always have foundation.
But distancing oneself from the opinion of the “experts” has an obvious risk in an increasingly polarized environment between black and white where the numerous shades of gray have disappeared.
In 2000, David Sackett published in the British Medical Journal the article “the sins of experts and proposals for their redemption”. He affirmed that experts prevent the advancement of science because those who present proposals different from the “official” ones are ignored or repudiated by the scientific-media officialdom. What happened with Laporte is but one example (yet another) of what Sackett expressed in his article, inviting all the experts in the world to resign and devote themselves to something else (as he demonstrated by his example) to favor the advancement of science.
“He’s sickly so much expert,
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1118019/
Synthesis
They pretend to make us believe that Joan Ramon Laporte “was” important in patient safety and pharmacovigilance, but that he is anchored in the “yellow card”, as if it were a defect to follow a line of work of half a century, from postal mail to the Eudravigilance database.
Their concerns about adverse effects and lack of efficacy of pandemic measures, including covid19 vaccines, are those of the world’s best professionals and scientists, such as John Ioannidis, Peter Gotzche, Alyson Pollock, Carl Henegan and many more; from Juan Erviti to Peter Doshi, passing through Tom Jefferson and a myriad of necessary dissenters, crushed by the unique irrational thought, by the barbaric healthism that favors business and inequality.
1 of 1
Update on the expert review of suspected psychiatric and sexual side effects to isotretinoin
Inbox
MHRA
3:35 PM (3 hours ago)
to me
Expert review of suspected psychiatric and sexual side effects to isotretinoin
View as a webpage / Share
Medicines and Healthcare products Regulatory Agency bulletin
Dear Stakeholder,
We would like to update you on our progress since the findings of the expert review by the Isotretinoin Expert Working Group (IEWG) were presented and discussed at the Commission on Human Medicines (CHM) in December.
It has been so valuable to involve patients and stakeholders throughout the review process, and we were very pleased so many of you chose to watch the open session of the CHM meeting.
We are now discussing the CHM advice with the companies that market isotretinoin in the UK. Unfortunately, we cannot give you a definite timeframe at the moment but as soon as these discussions are complete, we will be back in touch.
We very much want to involve patients and stakeholders in our ongoing work on isotretinoin and we will share more on how you can get involved when the recommendations are published.
If you require further information about this review, please email MHRACustomerServices@mhra.gov.uk.
Kind regards,
Patient, Public and Stakeholder Engagement team
The Romans had a word for the role Trudeau has assumed: dictator.
For two years now we have been screaming that the medical dictatorship is coming. Now it’s here:
https://www.bbc.com/news/world-us-canada-60381096
This is the nation that has long been the envy of the world.
My heart weeps.
If you don’t own your own body, you own nothing at all.
The principle of bodily integrity is simply too important to sacrifice for ANY reason — certainly not for an experimental gene therapy that was approved without any RCT data showing it lowered the rate of death or serious adverse events.
Trudeau and his billionaire paymasters and the rest of their obedient servants in government and the media have steadfastly refused to engage with us honestly on these issues.
Instead they have offered us shame, blame, character assassination, and relentless fear-mongering.
They have insulted our intelligence by offering us clowns, balloons, and free donuts, beer, and pot in exchange for getting the shot.
The mainstream media (which is heavily dependent on drug company advertising) has obediently parroted the party line.
The billionaire oligarchs who own the social media and whose fortunes have grown ever-more tumescent during the lockdowns have ruthlessly suppressed opposing viewpoints.
People fear losing their livelihoods for speaking out.
The truckers’ protest has been extraordinarily peaceful. The truckers themselves are losing hundreds of dollars a day while engaging in the protest.
Trudeau refused to engage with them honestly.
Instead he smeared them with the vilest invective.
When that didn’t work he brought in the police to steal their fuel and firewood, in the middle of the Canadian winter.
When that didn’t work he invoked a never-before-used act to award himself dictatorial powers.
The medical dictatorship we screamers have been warning everyone about for two years is here.
‘We are not using the Emergencies Act to call in the military. We are not limiting freedom of speech, freedom of assembly and people’s right to protest freely.’
Justin Trudeau@JustinTrudeau·
Officiel du gouvernement – Canada
I want to be very clear about what we are – and are not – doing by invoking the Emergencies Act, and how taking this step will help get the situation under control. In case you missed our announcement earlier today, watch this:
https://twitter.com/justintrudeau
Trudeau is accused of bringing ‘martial law’ to Canada
https://www.dailymail.co.uk/news/article-10513525/Trudeau-accused-bringing-martial-law-Canada-crack-Freedom-Convoy-Ottawa.html
Back at the Beginning
A Small Fringe Minority…
https://www.youtube.com/watch?v=cBp6KkHsZXs
That is using an elephant gun to kill a fly
https://www.youtube.com/watch?v=anK1k0gEgQU
He went through the whole phobe dictionary
https://www.youtube.com/watch?v=HQl1QXr7J78
it’s an extraordinary attempt to run a country…
……and then they came for our children. ‘Urges – children’ ?? There are threats against childrens’ rights to education unless vacced in some states. And this grossly disgusting network across countries is still in power 2 years after the beginning of a surge of powermongering lies and violence whether psychological or physical.
‘ The American Academy of Pediatrics (AAP) urges children and adults to get the COVID-19 vaccine and booster as soon as they are eligible’
James D. Campbell, MD, MS, FAAP
When can children get the COVID-19 vaccine?
. A COVID vaccine is available for children age 5 and up, and boosters are now authorized for those 12 years and older.
Vaccines for children age 6 months and up may be authorized next.
Clinical trials are underway for children age 6 months to under 5 years old.
‘Rmarkably effective and safe’ It;s not possible to know what the effects will be. just tell the truth and assist people to decide
Research shows these new vaccines are remarkably effective and safe. The American Academy of Pediatrics (AAP) urges children and adults to get the COVID-19 vaccine and booster as soon as they are eligible. Being vaccinated and boosted is especially important now with the rise in cases caused by the delta variant and omicron variants of the virus. More contagious strains can spread quickly and infect more children. Being fully vaccinated and boosted helps protect kids from serious disease and hospitalization from COVID.
Meanwhile Whitty in UK disengenuously disconnects himself from statements that vaccinations should be made compulsory by mandates for NHS workers , instead saying now it is a voluntary decision for them. thousands who declined the vaccine even after his fear mongering have lost employment
Sir Chris Whitty tells NHS medics they have a ‘professional…https://www.dailymail.co.uk › news › article-10488875
7 days ago — Sir Chris Whitty has written an open letter to NHS medics to tell them they have a ‘professional responsibility’ to get vaccinated against … Thanks sir Chris knight order of the bath (or some other nonsense)