This key post in the Politics of Care series stems from an invite from Norman Fenton to give a lecture on December 6 to a group interested in the evidence swirling around vaccines. It is accompanied by The Handmaid’s Vaccine on RxISK, which gives a video of the talk, whose text and slides are below. The sound effects in the video are slightly mixed at one or two point and you might need the text to clarify the points made.
https://www.realnotrare.com/post/davidhealy
This talk is for all who are interested in evidence and how we generate it as well as for a group of people who are pro-vaccine, to the point of being volunteers in clinical trials, but who have ended up being harmed by them. They are the ones doing the science and demonstrating what science means – as I’ll explain – but their work is written off as misinformation.
The company handling of SSRI harms shows we came to classify real Evidence as misinformation. Many of the company tricks involved in this effort to persuade us the world is flat will be known to you but the brazenness with which they were deployed and the failure of physicians to spot what was going on may be new.
Slide 2
I have a doctorate in Serotonin Reuptake and was keen to try the SSRIs early on. Two men I put on Prozac became suicidal. Their problems cleared after stopping and re-emerged on starting another serotonin reuptake inhibitor and stopped again after stopping. See Creaney et al.
This is as clear a causal connection as you can get anywhere in science. I sent the cases to Lilly for comment and presented them in forums. No-one offered alternate explanations. Others reported similar cases during the year to publication of this article.
Slide 3
This rash of cases forced FDA to require Lilly to defend their drug. As my article came out, Lilly published this Beasley et al article in the BMJ. It came out on the same day the company presented their case at an FDA hearing – September 20, 1991 – stating:
- The plural of anecdote is not data
- It’s the disease not the drug
- Are you going to believe the misinformation or the science?
- It’s all the fault of the Church of Scientology (1991’s Anti-vaxxers).
The BMJ article shows more suicidal events on Prozac but the paper said these were not statistically significant and so there was no problem. FDA talked about heart-breaking cases reported to them but concluded the science didn’t link Prozac to the problem.
Slide 4
Here is Tony Hill, who created RCTs, saying 20 years later RCTs can help assess one of the 100 things a drug does – something we might be able to use for treatment purposes. This, by definition, means RCTs are not a good way to evaluate a drug. See Clinical Trials are not Safe.
Saying RCTs are not the way the truth and the light, these days, is like saying the Bible, the Koran or the US Constitution aren’t reliable.
Slide 5 Watch this.
Slide 6
In a depression trial, investigators focus intensely on one thing – does Prozac have an effect on mood. Pretty well everything else is ignored. The statistics we use don’t work unless there is an intense effort to collect everything we can about this one outcome.
And so, depression trials miss something that happens to almost everyone who takes an SSRI within 30 minutes of the first pill – your genitals go numb. You can search the RCTs on these drugs and all you will find is that perhaps 5% of people have sexual issues on these drugs.
Emotional numbing is another extremely common effect almost completely missed. This is how these drugs help. This is how these drugs might help someone diagnosed as depressed but the key point is that it is much more common than depression recovery.
Similarly in the vaccine trials, the common thing is a multiplicity of Spike protein effects – doing this we hope might help but if we are hypnotized by what is hoped for we will miss and have missed what these Spike proteins are actually doing.
If we just depend on RCTs, we end up knowing almost nothing about a drug.
The idea that an RCT shows there is a favourable Risk-Benefit ratio for a drug or vaccine can only hold true if the thing we are looking at is the commonest thing this vaccine does – like a parachute for instance. The commonest thing is a life saved and the Risk Benefit is favourable but we don’t need an RCT for parachutes.
If what we are hoping for is pretty rare – as in vaccine or SSRI trials – and in particular if we don’t know what we are missing, such as an obliteration of our ability to make love, perhaps for all time, then claiming a favourable Risk Benefit ratio is psychotic.
Slide 7
The first rating scale for behaviour was the Hamilton Rating Scale for Depression. An aid to make sure physicians checked on most of the things that might be abnormal in depression while they were interviewing a patient. An aid to help a doctor do an interview that would help the patient to live the life they wanted to live.
If you cleave to the checklist you will do very standardized but possibly disastrous interviews. For instance, on the Hamilton Scale, there is an item on suicide which could stem from the illness or from the drug – it needs a judgement call as to which of these is responsible. Ditto for sex, for sleep. Just checking yes for suicidality risks going badly wrong.
Checklists like these however became viewed as scientific instruments. They look better to hospital managers than DH asking about your daughter or partner. Without judgements, in medicine we call these diagnoses, rating scales are meaningless – other than to help a doctor to help you to live the life Pfizer want you to live.
Slide 8
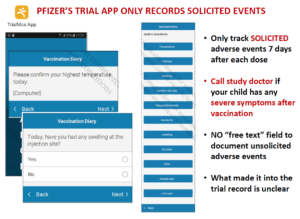
The latest twist on this story are the rating scales for adverse events companies now run electronically which let people rate up to 12 things that happen after the vaccine – such as sore arm, headache, nausea etc. This ensures certain events become statistically significant – and are put forward as a result as the only things we know for sure happen on the vaccine. See Johanna Ryan’s work on Virtual Trials.
There is no area for people to contribute anything else – so reports of other adverse events end up coming from outside the trial and are viewed anecdotes – misinformation. Companies like Pfizer tally anecdotes. What else would you do with misinformation?
Slide 9
Here is Fluvoxamine, an SSRI supposedly good for Covid. There have been lots of dropouts in the trials done on this, enough to invalidate the trial.
Side effects though could be endorsed on pre-populated lists that included cough, fever, nausea etc but not suicidality, homicidality, sexual dysfunction or the other things this drug causes that were likely responsible for the huge dropout rate.
Many look to drugs like this as an alternate to vaccines. Some doctors advocate them as our Hi Tech versus Albert Bourla’s Hi Tech. There are lots of low tech things that might be more helpful like getting you off some of the Tech you’re on rather than throwing more Hi Tech at you.
As we throw Hi Tech at you, we miss the fact that RCTs convert poisons, from whose use we hope to bring some good, into sacraments – something that can only do good. Most believers figure having as many sacraments as you can daily is a good thing where its seems equally obvious to most of us that taking more than one poison at the same time is unlikely to be all that safe.
One more quirk is companies always want their Ugly Ducklings to have an I’m a Swan moment – thalidomide was the fourth most profitable drug in the US last year but will be pushed down to fifth by Albertine this year. Thalidomide is a drug that causes suicidality, sexual dysfunction and birth defects – just like the SSRIs including fluvoxamine.
Slide 10
For drug companies, rating scales ensure you do an interview that produces figures which are the most seductive way to get the patient on their drug. The interview helps you to help them to live the life Pfizer want them to live.
This is not just true for rating scales, it is true for any measure – peak flow rates, bone densities, blood pressure or lipids, or sugar. It may be important to do something about some figures, but the goal is to help people to live the life they want to live – not the life Pfizer want them to live.
A stopwatch can be a wonderful motivator to achieve a dream – it provides data from one fraction of our lives. If we remain on top of that fine – but what about weighing scales? Just after they were introduced we got the first descriptions of anorexia nervosa. In the 1920s, they had norms for ideal weight attached to them and eating disorders mushroomed. They migrated into our homes in the 1960s and eating disorders became epidemic.
It’s very difficult to ignore figures for weight. Without data from every other aspect of our lives at the same time, we risk being trapped by this one data source. We become neurotic.
Can we let bone densities remain thin, or lipid levels remain high? Yes, we can. You think of post-mortems as something that reveal what we died from – they more often reveal what we can live with.
Slide 11
Figures create risks and pharma makes money from treating risks rather than diseases. We are seduced into taking drugs when we are healthy.
The Covid dashboards are a great mechanism to generate perceptions of risk and fear. The vaccines of course treat risks – not disease.
The Meatloaf title Paradise by the Dashboard Light is what Pfizer sees but its Hell by the Dashboard Light for us – this now extends to the evaluation of lectures and ensures we pander to people rather than challenge them.
It’s extraordinary how little we have put into treating SARS-Cov, the disease in this case and its associated pneumonia. Curing diseases is not a good business model.
Slide 12 As Peter Drucker, the Guru of Marketing Science put it 50 years ago:
The goal of marketing is to make selling superfluous. The aim is to know and understand the customer so well that the product or service fits and sells itself.
Slide 13
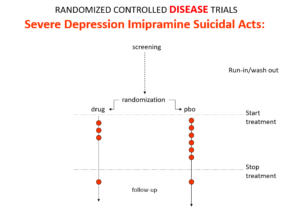
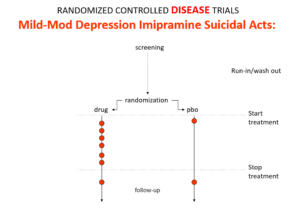
Imipramine was the first antidepressant. It beats the later SSRIs in RCTs. It treats melancholia – they can’t. They are useless for severe depression. Melancholia comes with a high risk of suicide.
Imipramine was launched in 1958. A year later at a meeting in England, Danish psychiatrists made it clear that while it was a wonderful treatment it made some people suicidal.
Let’s do a thought RCT of imipramine versus placebo in melancholia. Even though it can cause suicide, we would expect it to reduce the number of suicides because it treats the condition. This RCT would be great evidence antidepressants do not cause suicide.
Slide 14
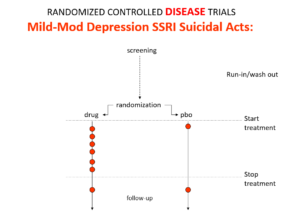
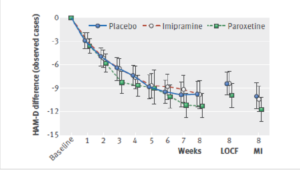
Here is the data on the trials in mild depression that brought the SSRIs to market – a doubling of suicidal events compared to placebo.
Slide 15
Imipramine looks the same in mild depressions. Now it too causes suicides. So RCTs tell us nothing about cause and effect – they can give us diametrically opposite answers. This is because these aren’t drug trials. They are Treatment Trials and in any clinical Trial, the condition confounds the effects of the drugs.
People evaluating drugs pre-RCTs knew this. When a patient becomes suicidal in a trial you have to use your judgement to work out what has happened but you are told not to.
Slide 16
This is true in every clinical situation where drugs and conditions cause superficially similar effects – diabetes and glitazones both cause heart failure, osteoporosis and bisphosphonates both cause fractures – and this makes it impossible for an algorithmic exercise as most RCTs are to establish what is happening.
Slide 17
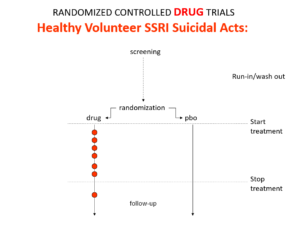
Here is what a real drug trial looks like. Companies ran these studies in the 1980s and found that SSRIs make healthy volunteers suicidal, cause dependence and sexual dysfunction but we heard nothing about this when the drugs launched. These Trials enabled companies to game their Treatment Trials to hide these problems.
Vaccine trials are healthy volunteer trials.
Slide 18
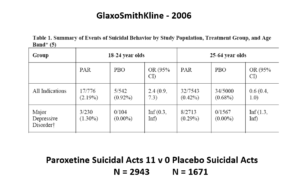
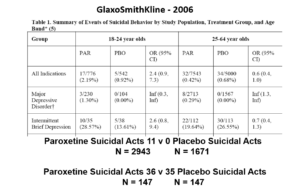
This slide shows data straight from a 2006 GlaxoSmithKline paper. GSK’s SSRI paroxetine was in trouble – the RCTs data for Major Depressive Disorder seem to show paroxetine causes suicidal events. The real data are likely worse that GSK admit to here.
Slide 19
But never fear RCTs come to the rescue. GSK also did trials in people with Intermittent Brief Depressive Disorders – IBDD. These are borderline personality disorder to most people – patients who have suicidal events much more often than anyone else. But these patients can meet criteria for depression and could be entered into Depression RCTs.
Slide 20
Prozac in these patients didn’t work. Paroxetine didn’t either and had a 3-fold higher suicidal act rate than placebo. GSK then did another trial in a similar group of patients. Why?
The answer is here. Here are IBDD data from the two GSK trials. I have seen other data for these two trials which make paroxetine look worse but let’s stick with GSK’s story. We could even add 16 more events to the paroxetine arm and still get the same magical outcome
When you add the IBDD data to the MDD data – all of a sudden paroxetine doesn’t cause suicidal events it protects against them.
Something like this must happen in every treatment trial with heterogenous patients – back pain, breast cancer, diabetes, hypertension, osteoporosis, parkinson’s disease. We can use an effect a drug causes to hide an effect a drug causes.
RCTs are not a way to work out what is going on. Back pain trials will insist you use analgesics rather than antibiotics – which is wrong for the 10% of backpains caused by infections.
Slide 21
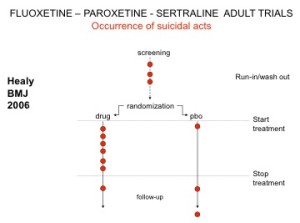
You’ve seen the son of this slide before. Here are the parents. In all AD RCTs there was a 2-week washout period during which patients were whipped off prior medicines. We now know this was a tricky thing to do – it gives lots of suicides – a bit like the two-week post vaccine period.
But companies argued as the patients were on nothing, all these events should be counted as placebo events – as the diagram here illustrates.
Slide 22
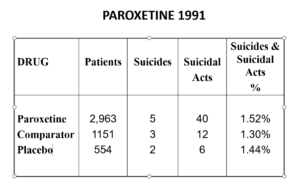
The Prozac 1991 paper had an increased number of suicidal events – but hey not statistically significant. Undo this maneuver –– and they are statistically significant.
Here are the paroxetine data presented to FDA. We’d prefer the figures for paroxetine to be better than placebo but what’s a fraction between friends.
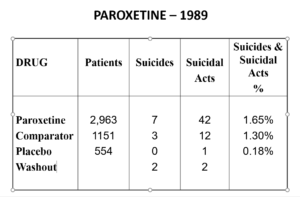
Slide 23
Undo the washout maneuver and this is what the data looked like. FDA knew what was going on and that it breached regulations and did nothing. And these figures don’t look like a drug that should be approved without warnings.
Slide 24
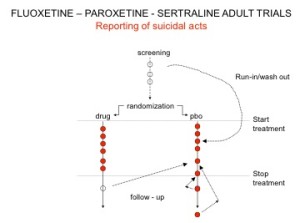
When that was rumbled, companies changed the game. Patients terminated from their SSRI who went into withdrawal and became suicidal were viewed as placebo, while those who stopped placebo and were put on an SSRI and committed suicide were classified as a placebo suicide – on an intention to treat basis. Regulators didn’t ask questions.
Slide 25
Sylvia Plath committed suicide a week into an antidepressant – a common timeframe.
Slide 26
This advert is for the type of antidepressant she was given, an MAOI, featuring a space shuttle, aimed to giving doctors the impression this drug will get their patients into orbit faster.
Slide 27
Here is a space shuttle – the safest transport ever per million miles travelled – but not so safe if expressed in terms of exits from and entries back into earth’s atmosphere.
Rather than express suicidal events per patient exposed companies stuck to events per thousand patient years – having taken care to ensure some patients doing well remained in extended follow-up for months or years.
Slide 28
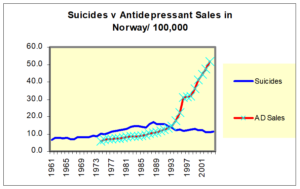
When the RCT data turned tricky and we got Black Box Warnings – companies turned to Real World Evidence – like national suicide rates. Here you see the claims for Norway which were typical of all Nordic countries – as SSRI use increased suicide rates fell – which is not compatible with the SSRI data.
If you look though suicide rates are going up with pre SSRI AD use until about 1988 – 3-4 years pre SSRI when they begin to fall.
Slide 29
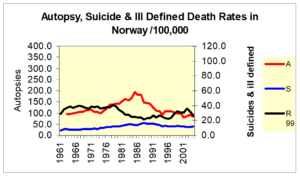
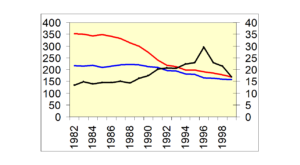
Here is Norway again where you see suicide rates falling from 88 or so and what you see if them rising as autopsy rates rise and then falling in step – as ill-defined deaths fall and rise.
Slide 30 This is true for all the Nordic countries – See Reseland et al.
Slide 31
We routinely hear that SSRI use is escalating. It’s not – the same number of people go on them each year. The increase speaks to the growing numbers who are dependent on them.
Slide 32
This has implications for suicide rates – you are only likely to see an effect on an index like this during the first few years. In the case of vaccines, this years rate of myocarditis and thromboses will become the new normal – See Healy and Aldred 2005.
Slide 33
In 1999 I was asked to participate as a speaking at a company symposium in London – speakers would need to produce articles for a supplement. I said yes and soon after had an email with my article. It was a great Healy article saying the things Healy say in the way he says them with Healy references. No one who knew my stuff would pick it out as not mine.
I emailed back saying I figured on writing my own. There was surprise at the other end but they said okay. I sent it on to them and they said this is rather good but there are some important commercial messages in the other one – we’ll get Siegfried Kasper to put his name on it.
Here it is – only one word changed from the original paper – the name of author Kasper. Everybody in Vienna knows this but its done no harm to SK’s career. You can find materials saying you can trust doctors like Kasper because they have written a 1000 articles or more. Its still a great Healy article etc.
Slide 34
A year later I was in Pfizer’s archive where even the loo-paper was stamped confidential. I came across this working document – which was the articles on Pfizer’s SSRI Zoloft being managed by Current Medical Directions – a medical writing company.
Slide 35
Inside there are pages listing the articles published or in train on Zoloft for the anxious, the depressed, the young, the old etc – here you see the PTSD page.
You will see on the right – the articles were written for these essentially negative studies saying the drug worked wonderfully well. One would go to NEJM – the other to JAMA. And on the left – you see TBD – authors names are To Be Determined. Pfizer’s marketing department will work out who would be the best sales people for the drug.
This is not just a mental health issue. It holds for all treatments across medicine.
Slide 36
Here is the most famous RCT of all time. It has a distinguished authorship line and is in the journal with the highest impact factor in child psychiatry and says paroxetine works wonderfully well and is entirely safe for children who are depressed.
Slide 37
This internal GSK document from 1998 shows the company knew the trial had shown the drug didn’t work and proposed taking out the good bits of the data and publishing those which is the article you have seen. New York State took a fraud action against GSK on this basis who were also fined $3 billion which led to access to the trial data and what you are going to see next.
Slide 38 The full story is on Study329.org
Slide 39
Through this legal action we got access to company data no one ever sees. The efficacy data is pretty irrelevant, but it was still possible to show that no matter which way you cut the data paroxetine was not more efficacious than placebo.
Slide 40
The tricks used to hide the problems were the real interest in these data.
The original article had 10 pages. Regulators see an 800-page Clinical Study Report (CSR) plus nearly 5000 pages of appendices – these are notional they are there but no-one in MHRA or FDA will look at them. We saw these and a further 77,000 Clinical Record Form (CRF) pages.
Point 2 points to data that just didn’t get transcribed from the 77000 pages to the 5000 pages. Point 10 is patients on placebo got SSRIs – I can explain how. But I want to focus on the coding issues.
Slide 41
The psychiatric adverse events all got grouped in CNS or neurological events – into which the groupers also put headaches and dizziness. The dizziness was not neurological – it was cardiovascular because it this case the comparator drug lowers BP especially when used in double or triple the adult dose.
The effect of this was to drown out the signal from psychiatric adverse events. So there is an issue about grouping. We were sensitized to this by Elizabeth Loder, the BMJ editor handling out paper – which took over a year to publish with 7 review rounds and 7 reviewers – who objected to our every mention of headache. As it turned out was a headache-ologist, who was an opinion leader for GSK but above all was the wife of an attorney working in Ropes and Gray who had been the lawyers defending GSK against the $3 billion fine.
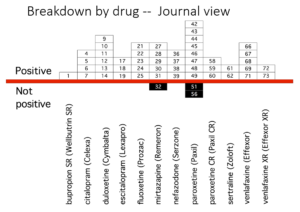
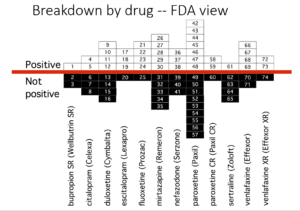
Slide 42
So here, leaving out headache and dizziness, in the lower bar you see the number of suicidal events in the Keller paper – once you decode them from emotional lability. In the middle bar, GSK revised this after being asked to do so by FDA when a fuss blew up. In the upper bar you see that we found more again – and there were more than we missed as I’m about to tell you.
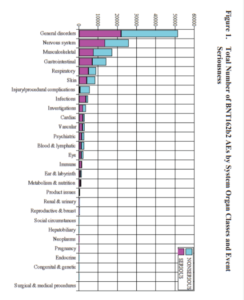
Slide 43
How does this fit Co-Vaccines? Well, here you see Pfizer’s report of their adverse event data – a ton of them have disappeared into a higher order coding group called General Disorders. The crimson half of the bar shows you these are serious, potentially lethal. General Disorders is a meaningless group – it needs unpacking.
Slide 44
In a Pfizer trial, one man poured petrol over himself and set a match to it, intending to kill himself. He died 5 days later from his burns. His death was coded as burns. But the company had to write a Serious Adverse Events narrative and if you got to see that you could work out that he should have been coded as suicide.
Slide 45
Companies have found a way to get around this, as found out after Study 329 finished. Here is a young man on a street waving a gun. Its Kyle Rittenhouse. In Study 329 a boy of 15 was picked up out on the street waving a gun around and threatening to kill people. He was hospitalized and should therefore have had an SAE narrative but the company coded him as intercurrent illness.
Four children dropped out of Study 329 coded as intercurrent illness – all were taking paroxetine. Add them into the picture you have just seen and things look a lot worse.
What is intercurrent illness? This was almost certainly an adverse event on paroxetine but invoking an intercurrent illness that means you really should not have been entered into this study means there is no need to write a narrative. This loophole has been there for 25 years and FDA have not moved to close it.
Slide 46
We know Astra-Zeneca broke the blind and got rid of serious adverse events like the ones that happened to Bri Dressen – see New England J of Misinformation. Here you see intercurrent illnesses turning up in this same Astra-Zeneca trial.
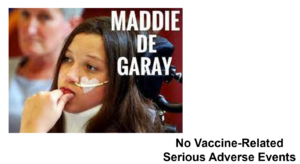
Slide 47
Here you have Maddie de Garay who has been tube fed and needs a wheelchair since a few days after the second dose of Pfizer’s vaccine in their trial for 12-15 year olds. But the company says no serious vaccine-related adverse events happened in this trial. They claim she has hysteria and of course that antedates the trial and so the vaccine can’t have caused it.
Slide 48
Few people know that FDA approved paroxetine for children – here is part of FDA’s 2002 letter of approval to GSK. The key bit is typed up so you can read it. The date is important – the Keller paper was 2001.
You can see here GSK told FDA that Study 329 was negative and you see that FDA agreed to approve the drug on the back of three negative studies and also agreed that there was no need to mention this in the labelling. Why would FDA do this?
Slide 49
Here are the published results of adult trials of antidepressants nearly a decade earlier. The picture looks pretty good.
Slide 50
But as Erick Turner has shown, this is how these studies looked to FDA. A different picture. Companies don’t leave negative studies unpublished, they know FDA are happy to let them publish negative studies as positive.
Why? Did GSK tell FDA – if you tell the world Study 329 was negative, we might get sued for fraud – which they did and fined for $3 billion. FDA don’t feel inclined to blow this whistle and MHRA and EMA have even less incentive.
Slide 51
So here is Study 329 again. The author is not listed. In the case of trials done in children, pretty well the entire literature was company written. The mismatch between what articles claim and the data when we see is the greatest known divide in medicine but likely not atypical. Study 329 was a good and ethical trial compared to some of the current vaccine trials.
There are now 45 negative trials for antidepressants in minors – out of 45 trials done. Yet antidepressants appear now to be the second most commonly taken drugs by teenage girls.
Slide 52
In this New England J of Misinformation article, the first thing to note is the author is not listed here. Twenty of the 29 apparent authors are company people. Few are clinicians and none are likely to have seen anyone harmed.
Second the trial was run by I Con rather than Pfizer who subcontracted to Palladium Research, who subcontracted to Ventavia and we know Ventavia ran a shit-show.
Anyone with experience of company trials knows that it is worth looking at the centres involved because for instance in a trial of aripiprazole where there might be 33 centres with 30 producing results for the drug that would not get approval, but perhaps 3 in places FDA won’t visit that found every patient put on the drug did fabulously and every on placebo was seriously injured or died a horrible death and adding both together produces a result that can squeak by FDA.
There is scope to wonder if something similar happened in this trial.
What we do know is that more people died on vaccine than placebo and lots more people disappeared on vaccine and FDA’s current leadership for whatever reason would prefer to be dead before anyone gets access to the data and their correspondence with Pfizer.
Slide 53
Is there a House in the Doctor? The medical drama House was watched and loved by many. Dr House was good at solving puzzling clinical cases by pulling on the thread of some minor detail which led to the answer.
Doctors today have close to lost the ability to say an evident X causes an evident Y – largely down to the mantra that only RCT evidence tells us what a treatment does and we can’t believe the evident anymore. Wife shoots husband point blank in chest – did she kill him? Who knows. In perhaps 1 case in 100 he had a heart attack just beforehand – we need to pass the 100 cases on to those experts in CDC, FDA, EMA etc and let them work it out.
As a result docs report maybe 1 in 10 or 1 in 100 serious side effects to regulators who file these away and do nothing with them.
This is unlike airline pilots who also report near misses and refuse to fly if these reports are not taken into account – after all if the customer dies the pilot does too. This is not true of doctors.
Slide 54
There is a profound misapprehension of the role of a regulator. They are not part of the health apparatus. Their job , perhaps easier to see in the case of food, is to decide if this yellow stuff is butter or lard colored to look like butter. If butter – it is not their job to decide if this is good butter or not or if butter is good for us or not. Ditto with drugs – the role is just to tick a box if certain criteria are met.
They have no abilities to or training in establishing if a drug or vaccine causes a problem.
Slide 55
Here is Walter Raleigh getting his head chopped off. After the fact legal systems recognized the injustice of convicting someone based on hearsay and said cases could only be decided on the basis of evidence in the room that can examined and cross-examined.
The first thing MHRA do with any reports is to remove the names of doctors and patients. This converts them into hearsay, anedcotes, misinformation. It means no-one can decide if there is a link.
MHRA will say till the crack of doom they are looking to find causal needles in the haystack of reports but faced with a needle-stack they can’t seem to spot a needle.
The key to determining cause and effect is an encounter between a doctor and patient. All the data is there. After a first run through there is a chance to follow up when oddities about the data come to mind. Remove the possibility for an ongoing two-way encounter like this and you remove the ability to establish cause and effect.
Slide 56
The one tool regulators have with anonymized drug reports is proportional reporting rates but as Matthew Crawford pointed out you can’t even use this for VAERS because you need lots of drugs in the mix for this to work properly. Besides proportional reporting rates are a cop-out. They might look more scientific than interviewing someone but they aren’t.
Slide 57
If someone commits suicide on an SSRI, their doctor will be advised by their insurer not to say the drug caused it or say anything that might lead to a further legal case. Insurance is supposed to be a business that supports us to take risks but is not doing this here.
If the doctor breaks ranks and blames the drug, a coroner, who can say a street drug caused a death, has no box to tick to implicate a prescription drug in a death.
Media guidance equally ensures journalists cannot say the obvious – the drug caused the suicide if the coroner hasn’t done so – and all this will apply to vaccines also.
If the coroner goes rogue and writes to the regulator and intimates that the evident cause of death was the drug or vaccine, the regulator will check on what the doctor has said and if the doctor didn’t finger the drug or vaccine – they won’t.
If the case is so Evident that both doctor and coroner go rogue, as in the Alana Cutland case, the regulator will respond, as MHRA did, that we only have a handful of reports like this – not enough to let us work out what might have gone on.
Slide 58
In the TV series House, the hero pays heed to tiny things that don’t fit the pattern and after twists and turns finds how it all hangs together.
When a wife shoots her husband in front of Dr House these days he seems unable to work out what’s gone on. Ok the guy may have had a heart attack at the same time and she shot him afterwards out of spite at being cheated out of a pleasant moment but 99 times out a 100 it’s pretty simple, she killed him. House though has lost this plot.
Some great doctors encourage their colleagues to report adverse events to regulators – which makes the problem worse. Regulators will file these reports away until the crack of doom. Unless doctors have the courage to say – look I know what I have seen and the vaccine or the drug killed my patient, they make things worse.
Dr House can’t get his head around the fact that with drugs and vaccines we hope to bring good out of the use of a poison, and sometimes people get poisoned. We and he prefer the idea he is giving sacraments – things that can only do good.
F Scott Fitzgerald once said that a sophisticated mind can hold two contradictory things in mind at the same time and still function – doctors could do this once but can’t now.
Science challenges Muslims and Jews, Xtians and Atheists to leave their biases at the door and come to a consensus about the data. But as in a jury trial, sticking with the data we still have to come to a verdict. A judgement. A diagnosis. It is not the role of a regulator to make diagnoses or deliver verdicts.
A verdict has effects in the real world just as much as shooting someone. There are evident effects from shooting on a husband who dies, and equally evident effects on the wife being tried.
This is important but more important for all of us just now are the effects on the doctor or failing to make any verdict, and just as much failing to make the diagnosis they know or suspect is the right one.
This failure transforms them into Model Doctors – a shrunken replica of the real thing.
When treating a patient, following the evidence can’t mean doing what ghostwritten fraudulent articles say. It has to mean following the person in front of me and coming to a consensus view just as a jury would.
If there is a mismatch between that and the so-called evidence – well all the books say that’s what moves science forward.
Slide 60
See The Handmaid’s Vaccine on RxISK – and its message about Albert and Ursula, the happy couple you see here.


































A “JUDGEMENT” Call … ‘Doctoring the Evidence’ …
AntiDepAware@AntiDepAware 1h
#WilfulBlindness
#Coroners
‘We blame doctors after our daughter killed herself while on acne drug’
“She was let down in life and she has been let down in death by the authorities’ steadfast refusal to recognise the role Isotretinoin played in her death.
“But we, the family and friends and the people who knew her know the real truth, and this will not be changed by the opinions of those who never met her – yet had the power to stand in judgment over her.”
“We know we will never get justice for our child, but we tried our best to get recognition for the role Isotretinoin played in Annabel’s death in the face of wilful ignorance by those who refuse to listen, we could do no more.
https://www.mirror.co.uk/news/uk-news/dangerous-acne-drug-caused-happy-25652833?utm_source=twitter.com&utm_medium=social&utm_campaign=sharebar
they will not hear about the true side effects including sudden suicidal impulses from those who glibly prescribe it.
Jonathan Leach said the evidence from experts on a potential link between Isotretinoin and self-harm was that there was ‘no settled and agreed view’.
https://www.dailymail.co.uk/news/article-10288549/Schoolgirls-suicide-not-directly-linked-acne-drug-Roaccutane-coroner-rules.html
Mr Wright had nothing to add to the statement, other than to say that he felt the whole way the case had been handled, including the inquest, had been ‘bizarre’.
Annabel Wright: Acne drug link to Ripon teen’s suicide ruled out
https://www.bbc.co.uk/news/uk-england-york-north-yorkshire-59568882
and that part of his evidence was outside of his area of expertise.”
Doctoring the Evidence: GlaxoSmithKline Pushes Depression Drug
Published by
Special to Corp Watch
By
Shelley Jofre
Monday, July 30, 2007
https://www.corpwatch.org/article/doctoring-evidence-glaxosmithkline-pushes-depression-drug
But the outcome of these the trials was not what GSK had been hoping for. Paroxetine proved no better than placebo. In the biggest trial, Study 329, which was conducted across several sites in the U.S., 11 of the 93 children who took paroxetine developed serious side-effects; seven had to be hospitalized. Significantly more had self-harmed or attempted suicide on the drug than on placebo.
The BBC (British Broadcasting Corporation) obtained confidential case reports from Study 329 which detail what happened to them:
This has been described by British psychiatrist Dr. David Healy as “the biggest medical scandal since thalidomide.” Dr. Healy, rather than the regulatory authorities charged with monitoring the safety of medicines, was instrumental in uncovering the evidence at the heart of the scandal. It was a series of U.S. legal actions that led Dr. Healy, as an expert witness, and the legal team with whom he was working, deep into GSK’s secret archives, where they found a series of damning internal documents.
‘This drug was GSK’s golden egg for many years and a lucrative business was built around its promotion. So it is perhaps not surprising the company dismissed my investigation as media scaremongering.
What is surprising, though, is how many doctors on both sides of the Atlantic who claim to practice evidence-based medicine still refuse to acknowledge the damage the drugs can do – even though the evidence comes from GSK’s own RCTs and the company’s internal correspondence clearly shows how they tried to cover it up. …
‘But results vary ‘
We are being treated like a herd of sheep harrassed by the dogs into a vaccine pen. How many of us can plough through this lot for example to make a decision and few will have access to someone to help us make a decision as best we can under the present circumstances And worse if we use the blogs and other research as part of a decision not to take the vaccine we are undermined in the same way reluctance to use SSRIs is undermined In the case of vaccines we can be coerced-sanctioned in the case of psych meds coerced-sectioned.
First studies suggest Omicron can evade vaccine protection, boosters needed
Variant spreading rapidly in Europe with case numbers doubling every three days or less.
INDIA-HEALTH-VIRUS
The Omicron variant can escape all the vaccines in use in Europe, but a third dose provides some protection
BY HELEN COLLIS
December 8, 2021 1:21 pm
First data from laboratory studies suggests the Omicron coronavirus variant can dodge the protection given by vaccines, although booster shots may help if enough doses can be administered quickly enough.
‘In the last 24 hours, study results from South Africa, Sweden and Germany, as well as from vaccine maker BioNTech, all suggest Omicron can “escape” the immunity gained via vaccination. But results vary….’ So .read on in POLITIC0
Ooops -the files have gone ‘missing’
‘European Commission President Ursula von der Leyen is under scrutiny over her text messages to the Pfizer CEO | Etienne Ansotte/European Union
BY LILI BAYER
September 17, 2021 9:59 am
European Ombudsman Emily O’Reilly wants to know more about European Commission President Ursula von der Leyen’s text messages with a pharmaceutical company chief.
In April, the New York Times reported von der Leyen had exchanged calls and texts with Pfizer Chief Executive Albert Bourla.
But when a request for access to the messages was submitted, the Commission said it had no record of them. Text messages, the Berlaymont argued, are generally “short-lived” and in principle excluded from its record-keeping. ‘
I think it is reasonable to have doubts when there was no warning that the vaccines would fizzle out after six months or so but ‘they’ made out that that was a possibility only after they started to wane. Did they know that would happen at the start of vaccine rollouts. Was it a case of nudging rather than a case of informing. Once vaccinated it seems reasonable to have another one just as a precaution. Did they know they might fizzle out without ‘boosters’. Once caught in a vacc trap it’s hard to say no to another one . and another one ..and another one. How much can a body take both biologically and psychologically. The pandemic has created a situation where refusal of vaccinations can lead for many to coercion-incarcerating and to avoid this involuntary consent (as not voluntarily given) to be vaccinated -just as refusal of psych meds can lead to coercion-incarceration-sectioning . Even though in both cases there are grounds to refuse . The for the common good is a powerful argument but the common good is not always to follow what politicians physicians scientists regulators uncle tom cobbly and all tell us. When so many have proven to be corrupted and/or our right to autonomy trashed by being pushed through regulations designed by those who are dehumanising people to a form of robot , they love themselves, love robots but not quirky human beings. There is no such thing as benign abuse but that is what is being claimed in the name of the common good
All4.com Homepage
A watered doen version on C4
Vaccine Wars: Truth About Pfizer: Dispatches
Pfizer’s Covid vaccine has saved millions of lives around the world. But with rising prices and record profits, is the drug firm profiteering from the pandemic?
No episode available to watch on demand on Channel 4
but might be able to download on Teleboy
Vaccine Wars – The Truth About Pfizer: Dispatches – Channel 4 – 10. December 2021, 20:30 – Teleboy Play
Record
This program is now only available in Replay.
Channel 4 Friday, 10. December • 20:30 – 21:00
Vaccine Wars – The Truth About Pfizer: Dispatches
Documentary
Pfizer’s Covid vaccine has saved the lives of millions around the world and the Government has chosen the Pfizer jab over the much cheaper Oxford AstraZeneca for its autumn roll-out of boosters. However, as Pfizer hikes up the price of its vaccine and enjoys record-breaking profits, questions are mounting over the pharmaceutical company’s behaviour. Reporter Antony Barnett considers whether its is unfairly profiteering from the pandemic, whether it has always played by the rules, and whether it is doing enough to stop people in poorer countries dying from Covid-19.
Our Response to Channel 4 Dispatches
Pfizer is disappointed to hear of the Channel 4 Dispatches documentary episode of 10th December 2021: “Vaccine Wars: Truth About Pfizer”.
The Dispatches documentary was being advertised before Pfizer itself was sent a list of areas to respond to with a very short window to reply.
We are concerned that unbalanced reporting will only serve to undermine the UK’s vaccination efforts at a time of much public concern. We have a number of concerns around specific accusations made in the approach to Pfizer, outlined below.
https://www.pfizer.co.uk/news-and-featured-stories/our-response-to-channel-4-dispatches
Vaccine Wars: Truth about Vaccines: Dispatches
https://www.channel4.com/programmes/vaccine-wars-truth-about-pfizer-dispatches/on-demand/73166-001
Pfizer’s Covid vaccine has saved the lives of millions around the world. But with rising prices and record profits, is the drug firm profiteering from the pandemic?
Home On website Real not Rare
Real Stories
Take Action
Contact
Videos
Groups
Other
Real Censorship
#RealNotRare published accounts with videos and presentations at event
Watch Now ‘What WAS your life like….’
Latest Videos
Latest Stories
Holly Lindberg
6 hours ago
Holly Lindberg
First Dose of Moderna on 03/12/21 Lot # 040A21A Second Dose of Moderna 04/09/21 Lot # 039A21A Blossom, Texas Q: What was your life like…
Amy Heffner
a day ago
3 min
Amy Heffner
First Dose of Pfizer on 09/09/21 Second Dose of Pfizer on 09/30/21 Texas Q: What was your life like before you got the vaccine? I was a…
Anthony Sanchelli
2 days ago
Anthony Sanchelli
First Dose of Pfizer on 04/25/21 Lot #EW0170 Second Dose of Pfizer on 05/16/21 Lot #EW0182 San Francisco, California Q: What was your…
Erin Van Vleet
3 days ago
Erin Van Vleet
First Dose of Pfizer on 01/18/21 Richmond, VA Q: What was your life like before you got the vaccine? My life before the vaccine was…
Michelle Johnson
4 days ago
Michelle Johnson
First Dose of Moderna on 01/13/21 Lot #012L20A Pennsylvania Q: What was your life like before you got the vaccine? Prior to vaccination I…
Mindy Brown
5 days ago
First Dose of Pfizer on 08/15/21 Georgia Q: What was your life like before you got the vaccine? I am a stay at home Mom so I basically…
Taylor Devereaux
5 days ago
Taylor Devereaux
First Dose of Pfizer on 09/10/21 Toronto, Ontario, Canada Q: What was your life like before you got the vaccine? I was a happy, 26 year…
Kirstie London
6 days ago
Kirstie London
First Dose of Pfizer on 05/19//21 Lot #EW0199 Second Dose of Pfizer on 06/30/21 Lot #FA9091 New Brunswick, Canada Q: What was your life…
SHARE YOUR STORY
READ MORE STORIES
“The FDA wants 55 years to release vaccine data”
Educate Yourself
FDA WANTS 55 YEARS:
https://www.reuters.com/legal/government/wait-what-fda-wants-55-years-process-foia-request-over-vaccine-data-2021-11-18/
The small amount of current data released, already leads to concerns:
https://www.react19.org/post/cumulative-analysis-of-early-on-pfizer-vaccine-data-released-by-the-fda-11-17
Lookup Your Reps
https://myreps.datamade.us/
Call Your Reps
Ask your representative to demand the FDA release the vaccine data in a timely manner.
The first 2.5 months of data is very concerning, and we have a right to complete disclosure and transparency.
Email Your Reps
Download this letter template for more talking points or to send your rep an email:
DOWNLOAD LETTER
Get Involved With Additional Actions Items >
Washington, D.C. , Nov 2nd, Event Presenters
Robert Kaplan, PhD
Stanford Robert M. Kaplan, PhD is currently a faculty member at the Stanford School of Medicine Clinical Excellence Research Center…
Iona Heath, CBE, FRCGP
Past President of UK Royal College of General Practitioners Inner city general practitioner in Kentish Town in London (1975-2010); now…
Peter Doshi, PhD
University of Maryland Peter Doshi, PhD, is an associate professor of pharmaceutical health services research at the University of…
Patrick Whelan, MD
University of California, Los Angeles Patrick Whelan MD Ph.D. is an Associate Clinical Professor of Pediatrics in the Division of…
Linda Wastila, BSPharm, MSPH, PhD
University of Maryland Linda Wastila, BSPharm, MSPH, PhD, is Professor and Parke-Davis Chair of Geriatric Pharmacotherapy in the…
Retsef Levi, PhD
MIT – Massachusetts Institute of Technology Retsef Levi PhD is the J. Spencer Standish (1945) Professor of Operations Management at the…
David Healy, MD, FRCPsych
McMaster University Dr David Healy is a Professor in the Department of Family Medicine at McMaster University in Ontario, having…
Aditi Bhargava, PhD
University of California, San Francisco Dr. Aditi Bhargava is a Professor in the Department of Obstetrics and Gynecology and the Center…
About Us
Our Purpose
Real lives are being affected by “not so rare” consequences.
Our purpose is to raise awareness of the many vaccine injuries that are happening, to create a community of public support, to give a voice to those who have been silenced, to have our elected officials acknowledge us, to encourage ways to advocate, and to change public perception. This is all in an effort to help the injured receive better healthcare and treatment. Vaccine injured individuals did their part by getting this vaccine, and now they need your help.
Although many do great with the vaccine, there is a chance for an adverse reaction. We believe where there is risk, there should be choice. Many are losing employment over these mandates, and we can not afford unemployment numbers to keep rising. We need to strengthen our economy; not lose our workforce.
This is not red vs. blue. This is not us vs. them. This is humanity vs. disease. #teamhumanity.
Our Demands
Our elected officials and federal health agencies must start acknowledging our adverse reactions to the vaccines. We have been silenced for nearly a year. Stop the censorship!
The medical community needs to be informed of these adverse reactions so that we can obtain better medical care.
Large scale research needs to be funded – to increase understanding of these reactions, find a path to recovery, and to help make the vaccines safer for everyone. There is currently no safety net for those Americans injured.
Stop the denial of religious and medical exemptions.
Stop the mandates. Where there is a risk – there should be a choice. We need to strengthen our economy; not lose our workforce.
We need the covid vaccines to be added under the VICP program, including eligibility for those who were injured under the EUA and clinical trials!
Getting the covid vaccine is a public service, and we rolled up our sleeves to do our part to stop the pandemic. It took bravery for those who got the vaccine early on or during the clinical trials. They willingly rolled up their sleeves and are now left out cold by the very same government who urged us to get our doses. This must change!
Who We Are
We are a completely independent, grass-roots movement comprised of people who have become united through our human suffering. This is not political. Many of us have lost loved ones to covid, and take it seriously. This is not about whether or not to vaccinate or wear a mask. This is about freedom of choice, informed consent, and quality healthcare for the injured.
1 of 1
ACTION ALERT! Take Action Against Tyrannical Federal Legislation
Inbox
Children’s Health Defense
Sat, Dec 11, 9:10 PM (8 hours ago)
Dear Susanne,
On November 30, H.R. 550, a federal bill passed in the U.S. House of Representatives.
The bill, “Immunization Infrastructure Modernization Act of 2021,” would expand state and local health department vaccine-tracking systems to monitor the vaccination status of American citizens. States would provide the information to the federal government.
The bill may be difficult to stop as it already has bipartisan support. It passed the House with the support of 294 U.S. Representatives, including all Democrats and 80 Republicans.
H.R. 550 is now under consideration by the U.S. Senate where, if passed, it could be implemented in under 12 months.
H.R. 550 would lead to a monumental invasion of our rights as American citizens. It would set an incredibly dangerous precedent and could lead to more vaccine mandates, and more restrictions of services and healthcare for the unvaccinated.
The bill also creates a mechanism for federal, state and local governments to enforce vaccine passports and possibly no-fly lists. And it would be costly to taxpayers — it appropriates $400 million dollars to expand vaccine tracking.
Part of this money would be spent on grants and cooperative agreements to state or local governmental entities that agree to adopt the new data collection guidelines set by the Centers for Disease Control and Prevention.
It’s imperative that we work to educate our Senators on why this legislation poses an unprecedented threat to freedom and liberty, and how it will lead to an unconstitutional invasion of privacy and loss of medical freedom.
Once freedoms are lost, they’re nearly impossible and extremely costly to regain through legal action. The time to act on this is now!
Please take a few moments to complete this action alert by simply entering your name and address, and a letter explaining why you oppose this legislation will automatically be sent to your federal representatives.
And mark your calendar now, to put aside time on Monday, December 13 to call your elected representatives. To make it easier, we’ve included a brief phone script. Calls to voice your concerns for any legislative matters are the most effective way to influence representatives, so please make the time to do this!
This is an invasion of privacy and freedom that no American citizen should be comfortable with and it’s critical that we all take action now! Thank you for taking the time to help us defend our freedoms and restore democracy.
Thank you,
The Children’s Health Defense Team
The Dispatches programme ‘Vaccine Wars’ is now being made available
free to play on All4. with e mail and password
Vaccine Wars: Truth About Pfizer: Dispatches
This intial message (above Dec 10th) has disappeared ie ‘No episode available to watch on demand on Channel 4
Vaccine Wars: Truth about Pfizer: Dispatches
https://www.channel4.com/programmes/vaccine-wars-truth-about-pfizer-dispatches/on-demand/73166-001
Pfizer’s Covid vaccine has saved the lives of millions around the world. But with rising prices and record profits, is the drug firm profiteering from the pandemic?
Rapid response to:
Evidence is insufficient to back mandatory NHS staff vaccination, says House of Lords committee
BMJ 2021; 375 doi: https://doi.org/10.1136/bmj.n2957 (Published 03 December 2021)
Cite this as: BMJ 2021;375:n2957
Evidence does not justify mandatory vaccines – everyone should have the right to informed choice
Dear Editor,
As doctors and health professionals, many of whom work in the NHS, we would like to express our opposition to anti-SarCov2 vaccination being mandated for any group of people, including health and care workers. We agree with the House of Lords committee that the evidence is insufficient to justify this measure, but the government and Parliament do not appear to be listening and mandatory vaccines for NHS staff looks likely to be passed into law this week.
We do not dispute that covid-19 can be and has been a dangerous infection, and we agree that vaccines are effective in many situations. However, there is considerable uncertainty about the effectiveness of the covid vaccines, some serious short-term complications and a lack of data on long-term harms. In this situation, it is imperative that people are able to make a fully-informed choice about whether to have the vaccine or not.
It is widely accepted that randomised controlled trials are the only means of providing robust data on the efficacy of medical interventions because observational data is subject to uncontrolled biases. Yet the randomised trials of the covid vaccines lasted for a very short time and were only powered to provide definitive statistical evidence on preventing ‘symptomatic infections’, not on preventing infection per se, hospitalisation or death. The trials also provided no data on whether the vaccines reduce transmission or not—things we have had to learn the hard way, through real world evidence like the rapid spread of the Delta and now Omicron variants.
Results from the randomised vaccine trials published so far suggested the vaccines were effective in reducing symptomatic infections for a few weeks. The average duration of follow-up for people in the first report from the Pfizer trial, on which licensing was based, was only 46 days, for example. [1] The recent report on data from people who had been in the trial for up to 6 months revealed that the mean total duration of follow-up for the primary outcome of the double-blind trial was 3.6 months for those who received the vaccine and 3.5 months for those allocated to placebo. [2] Moreover, only 7% of participants actually remained in the double blind trial for 6 months. [3] Real-world data are not consistent with the trial results, with high case numbers in doubly vaccinated individuals reported from the UK [4] and Israel [5], for example. This suggests either that effects of vaccines wear off quickly, and/or that some bias crept into original trial procedures, possibly due to unblinding caused by vaccine reactions [6] or other procedural irregularities. [7] The same observational data suggests the vaccines may reduce hospital admission and death due to covid infection, but, in the absence of data from randomised trials it is difficult to be certain, since unknown factors may bias the data in either direction.
More alarmingly, third and fourth ‘booster’ shots have not been tested in any randomised trials, and other data on the efficacy and safety of administering further doses are scanty.
In other words, data on the only outcome properly tested in randomised trials, the prevention of cases by two vaccinations, appear unreliable, possibly due to rapidly waning effects or other factors, and other outcomes and procedures have not been investigated in randomised trials, meaning there is no secure evidence either way.
As far as the safety of the vaccines is concerned, it is clear that rare but serious, and potentially fatal adverse effects occur, such as thrombosis and myocarditis, [8] and that these took months to identify. Long-term harms will be difficult to detect due to the short duration of the randomised trials, and will only become apparent in coming years.
There are also no data on groups who might be particularly adversely affected by the vaccine, such as those with, or at risk of autoimmune disorders, and there is little data on adverse effects of booster shots, which is significant since there have long been safety concerns about repeated exposure to mRNA technology. [9] Repeated booster vaccines therefore represent cumulative risk for untested benefit.
For young age groups, in whom covid-related morbidity and mortality is low, and for those who have had covid 19 infection already, and appear to have longstanding immunological memory, [10] the harms of taking a vaccine are almost certain to outweigh the benefits to the individual, and the goal of reducing transmission to other people at higher risk has not been demonstrated securely. [11]
Respecting people’s autonomy and bodily integrity is at the heart of human rights and medical ethics and the data currently available on the vaccines by no means justify over-riding these important principles. More good quality research and access to existing data from the vaccine trials are required for people to make fully-informed decisions about whether to take these vaccines or not. [12] Coercing people to have a covid vaccine, either through the threat of legal sanctions or, in the case of mandates for occupational groups, by depriving people of their livelihoods and careers, is not justified due to the prevailing uncertainty about the overall benefits of the vaccines, the unfavourable risk-benefit ratio for many groups, and, not least, the lack of data on long-term harms.
13 December 2021
Joanna Moncrieff
Professor of Critical and Social Psychiatry
Professor Peter C. Gøtzsche, Dr Rufus May, Dr Giovanni Dalla-Valle, Dr Rachel Brown, Dr Lili Dawson, Dr Andrew Docherty, Professor Gabor Keleman, Dr Brian Martindale, Dr John Mason, Dr Gad Meyer, Dr. Livia Hronska, Dr Tomasz Pierscionek, Dr Pino Pini, Dr Jessica Robinson, Dr Clive Sherlock, Dr Marianne Siapera, Dr Federico Soldano, Dr Maria Tsvetkova, Dr Cathy Wield, Dr Sami Timimi, Dr Kasha Siubka-Wood
UCL
@joannamoncrieff
There was a video on YouTube with this content but it is no longer available.
Maybe it would be better to upload it on the blog and and other platforms such as Bitchute that are being used by those who have real content.
Thank you for your work, as always.
Thanks for noting this
The You Tube video has been censored without them telling me why
I’d love to know what the problem with asking for the raw data might be
It is now available through the blog post and is on Rumble
https://rumble.com/vqua3j-where-does-the-misinformation-come-from.html
and
the Real not Rare site https://www.realnotrare.com/post/davidhealy
David
It took me almost a year to read your answer. I just did a post with the links.
Maybe my last post. Have a great week!
https://hellaheaven-ana.blogspot.com/
I hope its not the last post. It can seem difficult being up against something like this kind of censorship. But it needs people to think of a way around it – probably a smart way – and the more who are thinking about this the more likely we are to find one
D
Coronavirus (COVID-19)
Guidance and support
HomeHealth and social careMedicines, medical devicesPharmacyRegulatory approval of Pfizer/BioNTech vaccine for COVID-19
Medicines & Healthcare products
Regulatory Agency
Decision
Information for Healthcare Professionals on COVID-19 Vaccine Pfizer/BioNTech (Regulation 174)
Updated 14 December 2021
Contents
Regulation 174 Information for UK healthcare professionals
1. Name of the medicinal product
2. Qualitative and quantitative composition
3. Pharmaceutical form
4. Clinical particulars
5. Pharmacodynamic properties
6. Pharmaceutical particulars
7. Marketing authorisation holder
8. Marketing authorisation number(s)
9. Date of first authorisation/renewal of the authorisation
10. Date of revision of the text
Print this page
Regulation 174 Information for UK healthcare professionals
This medicinal product has been given authorisation for temporary supply by the UK Department of Health and Social Care and the Medicines & Healthcare products Regulatory Agency. It does not have a marketing authorisation, but this temporary authorisation grants permission for the medicine to be used for active immunisation to prevent COVID-19 disease caused by SARS-CoV-2 virus in individuals aged 12 years of age and over.
As with any new medicine in the UK, this product will be closely monitored to allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions. See section 4.8 for how to report adverse reactions.
1. Name of the medicinal product
COVID-19 mRNA Vaccine BNT162b2 concentrate for solution for injection
It does not have a marketing authorisation, but this temporary authorisation grants permission for the medicine to be used for active immunisation to prevent COVID-19 disease caused by SARS-CoV-2 virus in individuals aged 12 years of age and over.
I might have misunderstood as only caught snipett on news – but haven’t they started or going to start vaccinating children UNDER 12 yrs old…
There’s a lot of important very detailed in the web site but too much to post as a comment
All of Dr. Breggin’s YouTube videos were taken down after he interviewed me about my book on antidepressants. (The channel is still operating, but it contains only videos by others that he has linked to.)
The Medical Dictatorship is coming.
BTW, if anyone is interested in listening to the interview, it still can be found on Dr, Breggin’s website. Click on this link, and then scroll down to Episode # 492:
https://breggin.com/the-dr-breggin-hour-archives/?fbclid=IwAR0NbRThwjKDjEFrmBmgdTHQUzDM5PpYuwa7Xm-1K41Xd3Cv4D3fN55y-Dc
Right after I posted the above comment, I received a press release from Senator Johnson]s office. The US military is discharging active-duty service members who are refusing the covid shot — even the ones who have recovered from the covid and now have robust, long-lasting natural immunity.
They have stopped pretending this is about protecting our health.
Intended for healthcare professionals
News
Covid-19: “Most complex patient in the world” should have ventilation withdrawn
BMJ 2021; 374 doi: https://doi.org/10.1136/bmj.n2193 (Published 07 September 2021)
Cite this as: BMJ 2021;374:n2193
Read our latest coverage of the coronavirus pandemic
Clare Dyer
Author affiliations
A 56 year old woman described by doctors as “the most complex covid patient in the world” should have the ventilation keeping her alive withdrawn by the end of October, a High Court judge has ruled.1
Mr Justice Hayden, vice president of the Court of Protection, said it would be in her best interests to be moved from the intensive care unit to a quiet and private place where she could end her life surrounded by her close family members.
He said he had been told authoritatively that she was the most complex covid patient in the world, in terms of the neurological impact and complications of the disease. That conclusion had been arrived at following consideration of national and international case studies, he added.
The woman, referred to as AH, has been a patient at Addenbrooke’s Hospital in Cambridge since December 2020 and on a ventilator since January. Hayden said her clinical presentation was highly unusual and the impact of the illness had created “an invidious dilemma” both for her family and professionals.
AH has extensive brain damage and by March her peripheral motor nerves had little to no remaining function. She can communicate with her eyes and has some neck and lips movement, but no limb movement, and frequently exhibits distress.
She has four children and Hayden said some family members had been highly critical of the hospital. “At times, some of their behaviour has fallen below that which the nursing and medical staff should be expected to tolerate,” he added. He described the staff’s care of AH as “inspirational.”
Each family member had vacillated as to the right way forward, he said, and none of the available options were devoid of difficulty. Cambridge University Hospitals NHS Foundation Trust had applied to the court for a decision about AH’s best interests.
“It is an unusual feature of this case that while the way forward has been described as ‘finely balanced’ every single professional involved in AH’s care has reached the same conclusion—that continued ventilation is not in her best interests,” he said. During examinations, on every occasion, she has become distressed, cried, and appeared anguished.
As well as mechanical ventilation, she has a nasogastric tube, a urinary catheter, and a rectal tube to manage faeces. She requires frequent suctioning of her trachea to control respiratory secretions and is turned often to avoid pressure sores.
Hayden said the delicacy of the case arose from two important aspects. “AH is able to feel and show some degree of emotion. Predominantly, she now reveals pain and real distress. However, she plainly sustains comfort from the presence of her children who have been the focus of her life.”
He added, “The evidence, not least that given by the family, has identified a tentative plan which has crystallised, at least to some degree, during the hearing. As I have analysed, it is centred upon respecting AH’s dignity and promoting the best quality of life at this last stage. For it to be most effective it will require cooperation between the family and those caring for AH. This will require respect, each for the other. The time has come to give AH the peace that I consider she both wants and is entitled to.”
Continued ventilation had become burdensome and medically futile, he said, but it should be continued until all four children, including AH’s daughter in Australia, and other family members could be with her in a quiet place, which must be by the end of October.
“It provides an important opportunity for this close and loving family to be together at the end. The treating clinicians feel able to work with and perfect this plan and recognise that it is consistent with their own professional conclusions and reflective of the central importance of family in AH’s hierarchy of values and beliefs,” he concluded.
This article is made freely available for use in accordance with BMJ’s website terms and conditions for the duration of the covid-19 pandemic or until otherwise determined by BMJ. You may use, download and print the article for any lawful, non-commercial purpose (including text and data mining) provided that all copyright notices and trade marks are retained.
https://bmj.com/coronavirus/usage
Rapid response to:
Covid-19: Researcher blows the whistle on data integrity issues in Pfizer’s vaccine trial
BMJ 2021; 375 doi: https://doi.org/10.1136/bmj.n2635 (Published 02 November 2021)
Cite this as: BMJ 2021;375:n2635
Open letter from The BMJ to Mark Zuckerberg
Dear Mark Zuckerberg,
We are Fiona Godlee and Kamran Abbasi, editors of The BMJ, one of the world’s oldest and most influential general medical journals. We are writing to raise serious concerns about the “fact checking” being undertaken by third party providers on behalf of Facebook/Meta.
In September, a former employee of Ventavia, a contract research company helping carry out the main Pfizer covid-19 vaccine trial, began providing The BMJ with dozens of internal company documents, photos, audio recordings, and emails. These materials revealed a host of poor clinical trial research practices occurring at Ventavia that could impact data integrity and patient safety. We also discovered that, despite receiving a direct complaint about these problems over a year ago, the FDA did not inspect Ventavia’s trial sites.
The BMJ commissioned an investigative reporter to write up the story for our journal. The article was published on 2 November, following legal review, external peer review and subject to The BMJ’s usual high level editorial oversight and review.[1]
But from November 10, readers began reporting a variety of problems when trying to share our article. Some reported being unable to share it. Many others reported having their posts flagged with a warning about “Missing context … Independent fact-checkers say this information could mislead people.” Those trying to post the article were informed by Facebook that people who repeatedly share “false information” might have their posts moved lower in Facebook’s News Feed. Group administrators where the article was shared received messages from Facebook informing them that such posts were “partly false.”
Readers were directed to a “fact check” performed by a Facebook contractor named Lead Stories.[2]
We find the “fact check” performed by Lead Stories to be inaccurate, incompetent and irresponsible.
— It fails to provide any assertions of fact that The BMJ article got wrong
— It has a nonsensical title: “Fact Check: The British Medical Journal Did NOT Reveal Disqualifying And Ignored Reports Of Flaws In Pfizer COVID-19 Vaccine Trials”
— The first paragraph inaccurately labels The BMJ a “news blog”
— It contains a screenshot of our article with a stamp over it stating “Flaws Reviewed,” despite the Lead Stories article not identifying anything false or untrue in The BMJ article
— It published the story on its website under a URL that contains the phrase “hoax-alert”
We have contacted Lead Stories, but they refuse to change anything about their article or actions that have led to Facebook flagging our article.
We have also contacted Facebook directly, requesting immediate removal of the “fact checking” label and any link to the Lead Stories article, thereby allowing our readers to freely share the article on your platform.
There is also a wider concern that we wish to raise. We are aware that The BMJ is not the only high quality information provider to have been affected by the incompetence of Meta’s fact checking regime. To give one other example, we would highlight the treatment by Instagram (also owned by Meta) of Cochrane, the international provider of high quality systematic reviews of the medical evidence.[3] Rather than investing a proportion of Meta’s substantial profits to help ensure the accuracy of medical information shared through social media, you have apparently delegated responsibility to people incompetent in carrying out this crucial task. Fact checking has been a staple of good journalism for decades. What has happened in this instance should be of concern to anyone who values and relies on sources such as The BMJ.
We hope you will act swiftly: specifically to correct the error relating to The BMJ’s article and to review the processes that led to the error; and generally to reconsider your investment in and approach to fact checking overall.
Best wishes,
Fiona Godlee, editor in chief
Kamran Abbasi, incoming editor in chief
The BMJ
More members of the parliament and retired politicians are speaking out against increasing authoritarianism in UK but not enough -some of the hostility being ramped up in sections of the media against people who are declining vaccines and support for vaccine passports has a lot of public approval in UK
Wales Online
In full: Lord Frost’s resignation letter to Boris Johnson
PA Reporters & Will Maule 55 mins ago
Lord Frost has resigned as the UK’s chief Brexit negotiator, citing concerns about the Government’s “direction of travel”.
(Part of) The full text of Lord Frost’s resignation letter to the Prime Minister is below.
Dear Boris, (Prime Minister)
I have led our EU exit process for the two and half years since you became Prime Minister.
In those years we have restored the UK’s freedom and independence as a country and begun the process of building a new relationship with the EU.
You know my concerns about the current direction of travel. I hope we will move as fast as possible to where we need to get to: a lightly regulated, low-tax, entrepreneurial economy, at the cutting edge of modern science and economic change.
Three hundred years of history show that countries which take that route grow and prosper, and I am confident we will too.
We also need to learn to live with Covid and I know that is your instinct too.
You took a brave decision in July, against considerable opposition, to open up the country again.
Sadly it did not prove to be irreversible, as I wished, and believe you did too. I hope we can get back on track soon and not be tempted by the kind of coercive measures we have seen elsewhere.
Together we have put this country onto a new path. I am confident that under your leadership this newly free Britain can succeed and prosper hugely.
I wish you and the Government every success in that.
David