First child myocarditis ad I’ve ever seen pic.twitter.com/MtUx2PEqh3
— Anish Koka, MD (@anish_koka) September 17, 2022
Paul Pickerell
Imagine you were a healthy public-spirited 49 year-old in Texas. You volunteered to take part in Pfizer’s Covid Vaccine trial. Your medical history was just a touch of hay-fever. You went through all the check-ups to make sure you were eligible. You rolled up your sleeve on 29 August 2020. And had the first vaccine. You didn’t know whether it was vaccine or placebo. You go home feeling fine.
Seven days later you wake up with tightness across your chest and feel sick. You are rushed to the ER where an acute myocardial infarction (STEMI) is diagnosed. Heart Attack. You need four stents inserted into your coronary arteries.
The Principal Investigator logs this as a Serious Adverse Event. How serious? Grade 4 “life threatening or disabling adverse event”. (Grade 5 means you’re dead.)
Five days later you are discharged. You have now become a patient, on four types of medication.
You are said to have recovered but then find that you are excluded from further participation in the trial.
You may not have been told that you had been randomized to be given the BNT162b2 vaccine.
Then you read:
“In the opinion of the investigator, there was no reasonable possibility that the acute myocardial infarction was related to the study intervention, but rather it was related to undiagnosed obstructive coronary artery disease. Pfizer concurred with the investigator’s causality assessment.”
Seven days after the “study intervention”?
Disjointed thinking
This is the opinion of Principal Investigator Paul Keith Pickrell MD, a rheumatologist based in Austin, Texas at the Tekton Research Centre. Rheumatologists are into joints but what was PKP on when he wrote this? This is one of the most blatant pieces of disinformation in the Pfizergate files.
Mind you it could all be a joke as Pickrell, a native Texan with 28 years’ experience:
“believes that humor blended with professionalism is an important approach to establishing relationships with his patients”.
Flied Lice
Rhode Island is the smallest state in the US but has one of the longest coastlines. A 48 year-old cyclist from Rhode Island was another volunteer. Here is the Bike Path at Watchemoket Pond, Rhode Island.

Apart from a raised cholesterol for which he was taking a statin, this young man had no pertinent medical history. He took part in the Pfizer trial organised by Omega Medical Research in the southern suburbs of Providence, RI.
He received his second dose of BNT162b2 vaccine on 16 September 2020. Eleven days later whilst cycling, he fell off his bike and was found unresponsive. He had suffered a heart attack with ventricular fibrillation due to a STEMI myocardial infarction. He was fortunate to be resuscitated and was taken to hospital where coronary angiography showed his left anterior descending artery was completely blocked. A stent was inserted and he was discharged three days later on five additional medications. This too was classed as a Grade 4 Serious Adverse Event.
Omega Research is now part of the Velocity Clinical Research (which, however, has nothing to do with cycling).
Boss of Omega Research is Dr David L Fried, an internist at Coastal Medicine, who has acted as the Chief Principal Investigator on over 300 clinical trials. Omega was one of the select few trial centres that attracted a “non-routine” audit in October 2020. Fried will then have been grilled by Robert Cutler, the Director of Medical Quality Assurance at Pfizer.
David L Fried MD
In Dr Fried’s opinion there was no reasonable possibility that the acute myocardial infarction was related to the study intervention, concomitant medications, or clinical trial procedures, but rather it was related to cardiovascular risk. Pfizer concurred with the investigator’s causality assessment.
Here is another example of an investigator cooking the what? The cyclist surely figured the vaccine might have had a part to play in his near demise. Did he seek compensation from Pfizer? Can we find him – this man might have a lot to gain from being found.
Flied pulled in $1,345,828.14 from Pfizer Inc. in 2020. He looks rather prosperous in this promotional video for Theraworx to rub on your legs. Might have been a lot better for our cyclist than the vaccine.
Death Defying
Santa Fe is the area where the train-station is in downtown San Diego. A place where many homeless men hang out.
Subject C4591001 1152 11521497 was a 72-year-old white male. He had a pertinent medical history of type 2 diabetes mellitus (since 2010) and hypertension (since 2018). He was on metformin (since 2013) for diabetes, ibuprofen (since 2015) for osteoarthritis, lisinopril (since 2018) for hypertension, and doxazosin (since 2019) for benign prostatic hypertrophy.
He received Dose 1 of the Pfizer vaccine on October 7 2020. He reported syncope (fainting) on October 26, 19 days later. He missed Visit 2 on October 28 (Day 22). On November 6, the research team find out that he had been admitted to the hospital on Oct 26, the day of his ‘faint’. He fainted in the middle of the night. On admission he was transferred to the intensive care unit.
The subject tested negative for COVID-19 at the hospital. On an unspecified date, the syncope resolved and he was discharged from the hospital.
[Without a Diagnosis? Not possible. You don’t get admitted to ICU for fainting.]
On November 6 it was decided he would not get Dose 2 but he remained in the study to be evaluated for safety, immunogenicity, and efficacy.
During the follow-up, the site contacted the subject’s sister, who confirmed that he had died on November 11. The cause of death was reported as unknown. It was not reported if an autopsy was performed. A death certificate might be available at a later date.
In the opinion of the investigator, there was no reasonable possibility that the syncope was related to the study intervention, concomitant medications, or clinical trial procedures. Pfizer concurred with the investigator’s causality assessment.

The investigator for site 1152 was Don Brandon MD, CRF Trials in San Diego – “Dedicated to Excellence in Medical Research Since 1966”. He is also President of Altor Medicus, Inc. based in Santa Fe.
Dr Donald Milan Brandon is a native San Diegan. He was a member of the Alpha Epsilon Delta Pre-Medical Honor Society. He did clinical research at a local hospital facility for a year and then earned his M.D. degree at the University of Southern California. He is very much a family man. He is an avid sportsman and conversationist as well as hobbyist, enjoying everything from model trains to radio-controlled aircraft. Disneyland is his second home and he makes no excuses about it.
Brandon received “associated research funding” from Pfizer totalling $5,627,954.42 in 2021 and $3,252,213.90 in 2020.
Disappearing Death
The syncope was coded as a “Nervous system disorder”. Someone did some further investigation and found apparently that the man had shigella, which can cause diarrhea. But his death still does not appear.
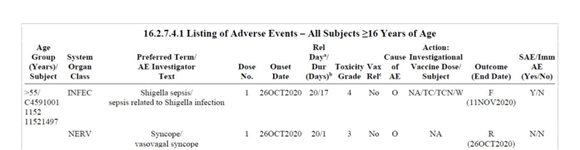
According to the narrative, sadly this volunteer died on 11 November, 36 days after his vaccination. No further details. This was just before the cut-off date for data submitted to the FDA and Polack’s NEJM paper. Had it been included, deaths in the vaccine arm would have increased by 50% (from 2 to 3). Is this why the clinical details are so vague?
Shigella was endemic in the homeless population of San Diego at that time. But there is nothing in this account or in the CRF that fits with this diagnosis.
By our calculations, there are at least 21 deaths in the vaccine arm and 16 in the placebo arm, many involving the heart, and it is likely there are even more waiting to be found, hidden under planks like codes for cardiac investigation,

TRUE!—nervous—very, very dreadfully nervous I had been and am; but why will you say that I am mad? The disease had sharpened my senses—not destroyed—not dulled them. Above all was the sense of hearing acute. I heard all things in the heaven and in the earth. I heard many things in hell. How, then, am I mad? Hearken! and observe how healthily—how calmly I can tell you the whole story…..
…
The officers were satisfied. My manner had convinced them. I was singularly at ease. They sat, and while I answered cheerily, they chatted of familiar things. But, ere long, I felt myself getting pale and wished them gone. My head ached, and I fancied a ringing in my ears: but still they sat and still chatted. The ringing became more distinct:—it continued and became more distinct: I talked more freely to get rid of the feeling: but it continued and gained definitiveness—until, at length, I found that the noise was not within my ears.
It grew louder…
“Villains!” I shrieked, “dissemble no more! I admit the deed!—tear up the planks!—here, here!—it is the beating of his hideous heart!”




New Zealand coroner just linked death in a relatively young man to myocarditis post vaccine.
https://www.rnz.co.nz/news/national/475109/man-s-death-ruled-a-result-of-rare-reaction-to-covid-19-vaccine
DH
Plandemic 3 – catch the rumble – it grew louder…
Pfizer concurred with the investigator’s causality assessment.”
Paul Keith Pickrell MD on Joints – Climbing Mount Everest – Thank God for every Tek and Patient that Participated
https://www.youtube.com/watch?v=FORYvFCQ0ZA&t=1s
‘ethically controlled’
https://www.youtube.com/watch?v=XTyFx0_Vzn4
Pfizer concurred with the investigator’s causality assessment.”
David L. Fried, M.D. On Recycling – Operation Warp Speed
New Novavax COVID-19 Vaccine Found To Be Safe and Effective in Trial – “Highly Efficacious and Very Safe”
https://scitechdaily.com/new-novavax-covid-19-vaccine-found-to-be-safe-and-effective-in-trial-highly-efficacious-and-very-safe/
Pfizer concurred with the investigator’s causality assessment.”
Donald Milan Brandon, MD on Vitals
https://www.vitals.com/doctors/Dr_Donald_Brandon.html
Pfizer Regulatory Quality Assurance
Sixteen (16) investigator site audits were performed between 29-Apr-2020 and 14-Nov-2020 for Study C4591001 Phase 2/3*.
https://phmpt.org/wp-content/uploads/2022/04/125742_S1_M5_5351_c4591001-fa-interim-audit-certificates.pdf#page=1
Polack, Fernando Pedro – Routine
Fried, David L. – Non Routine
Polack, Fernando Pedro – Non Routine
Teaser Plandemic 3
https://rumble.com/v1kdfd9-teaser-for-plandemic-3.html
Vax and a side of fries…
Comment from Johanna Ryan
The commercial trial sites that come up in these reports are among the highest-volume sites in Pfizer’s contract-research network. That’s kind of worrisome. So is the fact that many of them had unusually high “randomization” ratios: Well over 90% of the potential volunteers they screened got into the trial.
The California Research Foundation (CRF) screened 753 people and “randomized” 733 into the trial. That’s 97.3%, which those who know tell me is incredibly high. Michael Dever of CNS-Orlando enrolled 94.1% of the 882 folks he screened. And the very highest volume trial site, “Clinical Research Professionals” outside St. Louis, screened 942 people and “randomized” 934, which is a downright-freakish 99.1% success story.
CRF’s website fails to name a single physician (or even a single person!) in charge of its operations. However, it does include a portal for doctors and nurses to apply for work – either as freelance “investigators” (MDs) or employee-coordinators (RNs). It looks like a complex brokering arrangement involving an ever-shifting cast of (nameless) community doctors.
Clinical Research Professionals seems to operate in a similar way. It lists at least ten doctors, but they are all hyper-busy local specialists with really bustling practices. Currently the woods are full of CRO networks that tout their ability to rapidly turn any doctor’s office into a profitable research site, with minimal effort from the doctor. I wonder how many of those CRP doctors do actual research, and how many simply grant some nameless CRO access to their patients?
Mind you, none of these things are “proof” of corruption or data-doctoring. But they have to make you wonder: Who’s in charge at these research hubs, and who if anyone is watching over them?
It’s a question that rarely gets asked: Most Americans (reporters included) still believe that new drugs are “tested by the FDA” before approval. In reality, they are tested by big networks of gig-economy private researchers under the drug company’s control. The “results” the FDA gets to review are just the documents these private operators see fit to create, and release.
Who’s in charge at these research hubs, and who if anyone is watching over them?
Ventavia Research Group. Describing itself on its website as ‘the largest privately-owned clinical research company in Texas’,
Ventavia helped carry out the pivotal trial that was used to obtain authorization for Pfizer’s vaccine.
Company That Managed Pfizer Vaccine Trial Sites ‘Falsified Data’: Whistleblower
https://science.thewire.in/the-sciences/company-that-managed-pfizer-vaccine-trial-sites-falsified-data-whistleblower/
Pfizer has since hired Ventavia as a research subcontractor for four other vaccine clinical trials. These include trials on COVID-19 vaccines for children and young adults, pregnant women, and a booster dose.
—–
A researcher involved in the clinical trial that was carried out to obtain ‘emergency use authorization’ for Pfizer’s COVID-19 vaccine has revealed that some of the study data claimed to have been obtained from the tests were falsified. In an alarming investigation that has been largely ignored by most of the pharma-compliant mainstream media, the British Medical Journal (BMJ) reports that staff conducting quality control checks for the trial were ‘overwhelmed’ by the volume of problems they were finding. With the full data used to license Pfizer’s vaccine still not having been publicly released, the revelations seriously undermine the claim that the mRNA technology utilized in the injection has been scientifically ‘proven’ to be safe.
The accusations were made to the BMJ by Brook Jackson, who worked as a regional director for the Ventavia Research Group. Describing itself on its website as ‘the largest privately-owned clinical research company in Texas’,
Ventavia helped carry out the pivotal trial that was used to obtain authorization for Pfizer’s vaccine.
https://www.dr-rath-foundation.org/2021/11/whistleblower-reveals-use-of-falsified-data-in-pfizer-covid-19-vaccine-trial/
A disturbing list of concerns
Jackson’s concerns are anything but trivial. In addition to study data being falsified, she says vaccine packaging materials containing trial participants’ identification numbers were left out in the open at Ventavia, thus potentially threatening the ‘blinded’ status of the tests by making the interpretation of the results open to bias. In so-called ‘double-blinded’ studies, an approach generally considered to be the ‘gold standard’ in clinical research, neither the participants nor the researchers are supposed to know who is getting the drug/vaccine and who is getting the placebo. As such, for claims that Pfizer’s vaccine is ‘safe’ to be scientifically plausible, the ‘blinded’ status of the trial conducted to license it would need to be watertight and beyond reproach. Based on the BMJ’s investigation, it appears to fall short of this.
But the FDA is hardly alone in failing to fully investigate these allegations. By not reporting on the alleged use of falsified data in Pfizer’s COVID-19 vaccine trials, the majority of mainstream media outlets are similarly demonstrating that they too see the health interests of ordinary people as secondary to protecting pharma industry profits. With total global sales of COVID-19 vaccines forecast to reach as high as $190 billion this year, drug companies have a lot to lose when the coronavirus profit bubble finally bursts.
When the history of these times is eventually written, it is unlikely to look kindly on those who were trying to prevent this from happening.
INICIOMUSICACIENCIAECONOMIAAMBIENTEMÁS
HomeEconomiaSloppy Pfizer Booster Clinical Trial Consent Form Provides Way to Exclude Reactions That Require Emergency Care
ECONOMIA
Sloppy Pfizer Booster Clinical Trial Consent Form Provides Way To Exclude Reactions That Require Emergency Care
0
Share
Bloomberg Law complained recently that the consent forms for Covid 19 vaccine clinical trials are larded with unimportant information and difficult to understand. Based on our reading of a Pfizer consent form for a trial of a third shot of its Covid-19 vaccine, those aren’t the biggest causes for pause.
We’ve embedded a Pfizer consent form for a Covid-19 booster vaccine clinical trial below, which as of posting time was available at careidresearch.com. We strongly encourage you to read it in full.
We’ll discuss first how the form does not appear to have been reviewed by the oversight body tasked by the FDA to do so, and then will discuss why key parts are troubling.
The biggest issue, flagged in our headline, is that the consent form allows for participants who need emergency care and go straight to their doctor or hospital to be ejected from the study. But it’s not the only one.
Pfizer Consent Form Too Obviously Not Reviewed, Let Alone Negotiated, by FDA-Designated Overseer, the Institutional Review Board
It’s not hard to wonder if the sloppiness of this document is deliberate, that it’s the Big Pharma answer to Nigerian scam letter. As professor of information and spam expert Finn Brunton put it:
By making them really obviously fraudulent, you’re weeding out the skeptics. You’re only getting truly gullible responses.This consent letter looks design
ed to put off the legally savvy…who could be effective trouble-makers if anything went seriously wrong.
or as Pfizer which has committed numerous violations evidenced by documented fines
(see Violations Tracker) says – Important Notice
Beware of scams from individuals, organisations and Internet sites claiming to represent Pfizer in recruitment activities. A formal Pfizer recruitment process is required for all authorised positions posted by Pfizer prior to issuing an offer of employment. This Pfizer process includes an interview and never requires payment or fees from job applicants. If you receive a suspicious email message or phone call about recruiting on behalf of Pfizer, do not provide any personal information or pay any fees. Interested candidates should apply to current openings through this Pfizer website. Pfizer accepts no responsibility for any costs or charges incurred as a result of fraudulent activity.
“I think we should be deeply concerned,” said Lawrence Gostin, a Georgetown University professor who specializes in public-health law. “Putting it in a very crass way, money buys influence. And this is the worst kind of influence. Not just because it’s money — although that’s important, because money shouldn’t dictate policy — but also, because it’s preferential access, behind closed doors.”
Gostin said that such power, even if propelled by good intentions and expertise, is “anti-democratic, because it’s extraordinarily non-transparent, and opaque” and “leaves behind ordinary people, communities and civil society.”
While dozens of global health organizations participated in the world’s response to Covid, the POLITICO and WELT investigation focused on these four organizations because of their connections to one another — both Gavi and CEPI received seed funding from the Bill & Melinda Gates Foundation — and because they together played a critical role in advising governments and the WHO.
The WHO was crucial to the groups’ rise to power. All had longstanding ties to the global health body. The boards of both CEPI and Gavi have a specially designated WHO representative. There is also a revolving door between employment in the groups and work for the WHO: Former WHO employees now work at the Gates Foundation and CEPI; some, such as Chris Wolff, the deputy director of country partnerships at the Gates Foundation, occupy important positions.
Much of the groups’ clout with the WHO stems simply from money. Since the start of the pandemic in 2020, the Gates Foundation, Gavi, and the Wellcome Trust have donated collectively more than $1.4 billion to the WHO — a significantly greater amount than most other official member states, including the United States and the European Commission, according to data provided by the WHO.
“You have to remember that when you’re dealing with the Gates Foundation, it’s almost like you’re dealing with another major country in terms of their donations to these global health organizations,” a former senior U.S. health official said.”
“Now, the four groups are spending millions of dollars to lobby the U.S. and EU to embrace their priorities for the next pandemic, some of which include strengthening local health systems. Other initiatives involve investing more money in research and development, hoping to create next-generation vaccines and expanding surveillance networks across the globe.”
“No one’s actually holding these actors to account,” said Sophie Harman, professor of international politics at Queen Mary University of London. “And they’re the ones that are really shaping our ability to respond to pandemics.”
https://www.politico.com/news/2022/09/14/global-covid-pandemic-response-bill-gates-partners-00053969
“money buys influence” is precisely correct. The slush pots of money swirling around the medical system creates a tremendous moral hazard. Close your eyes to the truth and keep your lovely house and nice cars. Say something and lose it all.
Money doesn’t really do as much as you may think. People don’t usually change views because they are paid. The people with views that drugs and vaccines can do no harm are more likely to be paid and have their views disseminated than those who figure of course its just common sense that drugs and vaccines while helpful can also harm.
In the case of the vaccines some of the people I most respect have been pro-vaccine and are not willing to talk about harms or support me in talking about harms – without being paid a cent by anyone.
So which is the bigger factor – money or belief systems? Money doesn’t buy belief. The beliefs link to black and white thinking that sees the vaccines or drugs as good and not something to allow be others to blaspheme against.
David
A person with a strong belief will not be swayed by listening to the other side of the argument. A person attracted by the money is a person whose ‘belief system’ is already weak. That type of person will sway in whichever way provides them with the best outcomes. For that very reason, they cannot bear the idea of listening to anyone who has a different belief – they just can’t cope with the idea that someone else’s idea may question their own stand on a subject. This often leads to an attempt at humiliating the strong believer which we see in the media almost daily.
What’s Not Being Said About Pfizer Coronavirus Vaccine
https://journal-neo.org/2020/11/13/what-s-not-being-said-about-pfizer-coronavirus-vaccine/
Bill Gates is actively financing and promoting new untested vaccines supposed to keep us at least somewhat safe from a ghastly death from the novel coronavirus and supposedly allow us to resume somewhat “normal” lives. The Pharma giant Pfizer has now announced what they claim were spectacular results in initial human tests. They use an experimental technology known as gene editing, specifically mRNA gene-editing, something never before used in vaccines. Before we rush to get jabbed in hopes of some immunity, we should know more about the radical experimental technology and its lack of precision.
The financial world went ballistic on November 9 when the pharma giant Pfizer and its German partner, BioNTech, announced in a company press release that it had developed a vaccine for Covid19 that was “90%” effective.
Because of the degree of global panic engendered by WHO over the coronavirus, caution is thrown to the wind. Vaccine makers all have legal indemnity, meaning they can’t be sued if people die or are maimed from the new vaccine.
If that should happen in a major human trial the outcome could be disastrous. This serious adverse effect may not even be detected by a clinical trial especially in highly biased clinical trials laden with conflicts of interest involving vaccine companies. Even when a serious adverse event is detected, this is usually swept under the rug.”
Clearly the well-established Precautionary Principle–if in serious doubt, don’t– is being ignored …
F. William Engdahl is strategic risk consultant and lecturer, he holds a degree in politics from Princeton University and is a best-selling author on oil and geopolitics, exclusively for the online magazine “New Eastern Outlook”.
Well-qualified links, included…
Open Payments Data – CMShttps://openpaymentsdata.cms.gov
Search Open Payments. The Open Payments Search Tool is used to search payments made by drug and medical device companies to physicians, physician
An official website of the United States government
Information
PFIZER INC.
NY United States
2021
Payment type
General payments
Recipient type
All Recipient Types
Recipient
Nature of payment
All Natures of payment
Totals by payment type in 2021
What are the different payment types?
Briefly
https://openpaymentsdata.cms.gov/
Here you can find what any investigator was paid by any drug company
Associated Research is: Funding for a research project of study where the physician is named as a principal investigator
General Payment is: Payments that are not associated with a research study.
Research Payment is: Payments that are associated with a research study.
Not very clear is it?
Dr Naomi Wolf and Amy Kelly: What did the hundreds of thousands
of Pfizer documents from the FDA expose?
Video – info 10mins in
https://dailyclout.io/amps-reclaiming-medicine-conference-featuring-dr-naomi-wolf-and-amy-kelly/
“More people have been dying this year due to irregular heartbeat official figures suggest”
“Sarah Caul, ONS head of mortality analysis, said the figure had been “driven by higher than expected numbers of deaths since March, which could be caused by a combination of factors.
“Across March, April and May we saw increases in deaths due to cardiac arrhythmias, predominantly among those aged 80 and above.
“Total deaths registered in England and Wales were 10.3% below average in January and 7.3% below in February, but were above average in every month from March to June.
“Further work needs to be done to understand any link between the long-term effects of Covid and increasing cardiac deaths.”
https://news.sky.com/story/irregular-heartbeat-likely-to-be-one-of-the-reasons-more-people-than-usual-have-been-dying-this-year-12701948
Wonder what the numbers are for prescription of Citalopram and EScitalopram – prolongation of QT interval – as well as the vaccine during this period.
I am not allowed to copy more than this but there are named lead investigators and others who might be willing to answer a few of the questions Pfizer blocks with a standard letter.
…
CORONAVIRUSAugust 13, 2021
UC Davis School of Medicine partners with Pfizer to study COVID-19 booster vaccine
60 participants will help determine if third shot can protect against Delta variant
(SACRAMENTO)
As COVID-19 booster shots begin for those with compromised immune systems, researchers at the UC Davis School of Medicine continue to collect data from clinical trial participants. The goal is to help federal health authorities decide whether to expand vaccine booster shots to more people.
UC Davis is one of only 150 sites taking part in the major clinical trial that led to the federal government’s Emergency Use Authorization (EUA) of the widely used Pfizer COVID-19 vaccine.
UC Davis is one of only 150 sites taking part in the major clinical trial that led to the federal government’s Emergency Use Authorization (EUA) of the widely used Pfizer COVID-19 vaccine.
(I realise this is old news but with actual names it might be possible to bypass Pfizer
Haczku said the data collected by participants is processed by Pfizer, which then shares its results with the federal government. The ongoing worldwide trial involves 40,000 participants. They were the first to receive COVID-19 vaccines, several months before the U.S. Food and Drug Administration approved the EUA for the vaccine developed by Pfizer Inc. and BioNTech in December 2020.
Q how is the data processed by Pfizer?
Nicholeth Santiago also accepted the call to participate, without hesitation.
She happens to know a lot about research – she works at the UC Davis Medical Center helping to recruit and coordinate participants for studies in the Department of Emergency Medicine.
Q Considering the criticisms of the consent process, can we have a copy of the one they use?
Angela Haczku, co-principal investigator, said Pfizer was impressed with the “efficiency of trial participant recruiting, quality of data collection, recording and timeliness of submission, and professionalism of our coordinators and physicians led by Dr. Tim Albertson,” who is the principal investigator.
Q can we see the details of how this this impressive work was carried out so efficiently?
UC Davis is one of only 150 sites taking part in the major clinical trial that led to the federal government’s Emergency Use Authorization (EUA) of the widely used Pfizer COVID-19 vaccine.
Q How was the data collected from all sites and did pfizer do the final collation from all 150 sites?
Yaffa Shir-Raz
@YaffaRaz
Health and risk communication researcher. Health journalist.
GB News and the wonderful Neil Oliver did an important follow up tonight on the #IsraeLeak story we broke a month ago. Not only has the Moh kept a deafening silence ever since, but they pulled the plug on the study, knowing full well the implications.
https://twitter.com/YaffaRaz
Brook Jackson Retweeted
Efrat Fenigson
@efenigson
·
Part 2 –
Efrat Fenigson
@efenigson
· 13h
2/ Tonight on @GBNews: Israel Pfizer Leak with Dr. Yaffa Shir Raz. Israeli Health Ministry knows pfizer jabs are 1) causing long-term adverse effects, 2) introducing “re-challenge”: adverse effects repeat with each dose, 3) exposing them to liability
@YaffaRaz @thecoastguy
Neil Oliver Retweeted
Efrat Fenigson
@efenigson
Part 3 –
Efrat Fenigson
@efenigson
· 13h
3/ Tonight on @GBNews: Israel Pfizer Leak, interviewing Will Jones of @LD_Sceptics about the censorship & lack of media coverage for this huge headline story about the Israel – the world’s lab – and the hiding of this truthbomb.
@YaffaRaz @thecoastguy
https://twitter.com/thecoastguy
Albert Bourla
@AlbertBourla
·
14h
I have tested positive for COVID. I’m feeling well & symptom free. I’ve not had the new bivalent booster yet, as I was following CDC guidelines to wait 3 months since my previous COVID case which was back in mid-August. While we’ve made great progress, the virus is still with us.
Science will Win #Pfizer…
In the area of product safety, Pfizer’s biggest scandal involved defective heart valves sold by its Shiley subsidiary that led to the deaths of more than 100 people. During the investigation of the matter, information came to light suggesting that the company had deliberately misled regulators about the hazards.
The Food and Drug Administration expressed greater concern about reports of dozens of fatalities linked to heart valves made by Pfizer’s Shiley division. In 1986, as the death toll reached 125, Pfizer ended production of all models of the valves. Yet by that point they were implanted in tens of thousands of people, who worried that the devices could fracture and fail at any moment.
In 1991 an FDA task force charged that Shiley had withheld information about safety problems from regulators in order to get initial approval for its valves and that the company continued to keep the FDA in the dark. A November 7, 1991 investigation in the Wall Street Journal asserted that Shiley had been deliberately falsifying manufacturing records relating to valve fractures.
Faced with this growing scandal, Pfizer announced that it would spend up to $205 million to settle the tens of thousands of valve lawsuits that had been filed against it. Even so, Pfizer resisted complying with an FDA order that it notify patients of new findings that there was a greater risk of fatal fractures in those who had the valve installed before the age of 50. In 1994 the company agreed to pay $10.75 million to settle Justice Department charges that it lied to regulators in seeking approval for the valves; it also agreed to pay $9 million to monitor valve patients at Veterans Administration hospitals or pay for removal of the device.
Christine Dolan interviews Sasha Latypova and Attorney Warner Mendenhall
https://creativedestructionmedia.com/video/2022/09/17/livestream-sunday-1230pm-est-the-globalists-in-plain-sight-8/
“This show is about evidence today”
Chris’s link is good value for money and well worth watching. Get’s better as it goes on.
Warner Mendenhall as usual is fun.
He refers to the Corporate Integrity Agreement between the Office of Inspector General of the Department of Health and Human Services and Pfizer Inc. which runs until May 2023.
Pfizer agrees not to do Fraud.
https://oig.hhs.gov/fraud/cia/agreements/Pfizer_Inc_05232018.pdf
Pfizer agrees not to do Fraud effect
https://www.youtube.com/watch?v=xDuZGxhMQls
Strange – there’s a lot of people we are aware of who should be on this list. What is the relationship of OIG with you know who..
An official website of the United States government Here’s how you know
HHS Office of Inspector General
MENU
Exclusions Program
OIG has the authority to exclude individuals and entities from Federally funded health care programs.
Working with Federal and State Partners
This webpage provides information about OIG’s exclusion authority and activities. OIG has the authority to exclude individuals and entities from Federally funded health care programs for a variety of reasons, including a conviction for Medicare or Medicaid fraud. Those that are excluded can receive no payment from Federal healthcare programs for any items or services they furnish, order, or prescribe. This includes those that provide health benefits funded directly or indirectly by the United States (other than the Federal Employees Health Benefits Plan).
OIG maintains a list of all currently excluded individuals and entities called the List of Excluded Individuals/Entities (LEIE). Anyone who hires an individual or entity on the LEIE may be subject to civil monetary penalties (CMP). To avoid CMP liability, health care entities should routinely check the list to ensure that new hires and current employees are not on it.
Dr Aseem Malhotra
@DrAseemMalhotra
·
16m
BREAKING: ‘We have a psychopathic entity influencing health policy and that needs to stop.
It needs to stop now’
Dr Aseem Malhotra
@DrAseemMalhotra
THIS is at the root of our healthcare crisis. The paper below highlights just how corrupted the system has become by psychopathic entities
Dr Aseem Malhotra
@DrAseemMalhotra
I’ve spend 9 months on this paper and today it’s finally published after rigorous and long peer review process. It’s perhaps the most important work of my career so far and it has implications for every human being on the planet.
https://insulinresistance.org/index.php/jir/article/view/71/224
https://insulinresistance.org/index.php/jir/article/view/72/228
Download these PDF Files
https://twitter.com/DrAseemMalhotra
Dr Aseem Malhotra Retweeted
Josh Guetzkow
@joshg99
·
2h
Replying to @DrAseemMalhotra @DoctorTro and 9 others
Congrats and thank you! Literally one of the most important articles of the resistance!
Suspend All COVID-19 mRNA Vaccines Until Side-Effects are Fully Investigated, Says Leading Doctor Who Promoted Them on TV
BY WILL JONES
25 SEPTEMBER 2022
https://dailysceptic.org/2022/09/25/suspend-all-covid-19-mrna-vaccines-until-side-effects-are-fully-investigated-says-leading-doctor-who-promoted-them-on-tv/
Leading doctors have joined a call to suspend all COVID-19 mRNA vaccines until serious side-effects are fully investigated and the raw trial data from Pfizer’s COVID-19 vaccine trial are released for independent analysis to help determine the true benefits and potential harms for different age groups.
Writing in the peer-reviewed Journal of Insulin Resistance, one of the U.K.’s most eminent Consultant Cardiologists Dr. Aseem Malhotra, who was one of the first to take two doses of the vaccine and promote it on Good Morning Britain on TV, says that since the rollout of the vaccine the evidence of its effectiveness and true rates of adverse events has changed.
In a two-part research paper entitled “Curing the pandemic of misinformation on COVID-19 mRNA vaccines through real evidence-based medicine”, real-world data reveal that in the non-elderly population the number needed to vaccinate to prevent one death from COVID-19 runs into thousands and that re-analysis of randomised controlled trial data (that first led to approval of the vaccines for Pfizer and Moderna) suggests a greater risk of suffering a serious adverse event from the vaccine than being hospitalised with COVID-19.
Dr Aseem Malhotra
@DrAseemMalhotra
And this was for more lethal Delta variant. Wait until I present the data for omicron tomorrow at press conference. People will be SHOCKED
https://www.corp-research.org/pfizer#:~:text=Search,Corporate%20Rap%20Sheet
Pfizer: Corporate Rap Sheet
In 2009 Pfizer had to enter into a Corporate Integrity Agreement with the Inspector General of the Department of Health and Human Services. ( Seems this is no longer on the web )
9 Years later after many gross infringements they were obliged to enter an Agreement which was signed by binding signatures for purposes of this CIA.
Corporate Integrity AgreementCORPORATE INTEGRITY AGREEMENT
BETWEEN THE
OFFICE OF INSPECTOR GENERAL
OF THE
DEPARTMENT OF HEALTH AND HUMAN SERVICES
AND
PFIZER INC.
I. PREAMBLE
Pfizer Inc. (Pfizer) hereby enters into this Corporate Integrity Agreement (CIA)
with the Office of Inspector General (OIG) of the United States Department of Health
and Human Services (HHS) to promote compliance with the statutes, regulations, and
written directives of Medicare, Medicaid, and all other Federal health care programs (as
defined in 42 U.S.C. § 1320a-7b(f)) (Federal health care program requirements).
Contemporaneously with this CIA, Pfizer is entering into a Settlement Agreement with
the United States……….a VERY long document
ON BEHALF OF PFIZER INC.
___/Rady Johnson/_________________
RADY JOHNSON
Executive Vice President
and Chief Compliance and Risk Officer
Pfizer Inc.
____May 21, 2018____________
DATE
____/John Rah/____________________
JOHN RAH
Counsel for Pfizer Inc.
DLA Piper
____May 22, 2018____________
DATE
___/Joshua Levy/___________________
JOSHUA LEVY
Counsel for Pfizer Inc.
Ropes & Gray CORPORATE INTEGRITY AGREEMENT
(Ropes and Gray rings a bell?
Since 2018 Pfizer has been fined for more gross infringements re violation tracker.. But their vaccines are being used again in boosters currently in UK
Ding dong Ropes and Gray above, rang a bell Here it is.
Dr. David Healy
Psychiatrist. Psychopharmacologist. Scientist. Author.
Mark and Barb and Eric and Carole on David Healy Blog
May, 30, 2022 | 13 Comments
Born in the USA
Multi-Regional Clinical Trials (MRCT) was born in the USA in 2009. Pfizer had just resolved a case with the US Department of Justice for $2.3 billion, then one of the biggest resolutions of an action against a corporation.
The company convened a meeting of other pharmaceutical companies, contract research organizations (CROs), institutional review boards (IRBs) (ethics committees in Europe) and lawyers, especially Ropes and Gray, a Boston law firm, later involved in GSK’s defense against a Department of Justice action, resolved in 2012 with a payment of $3 billion. This meeting created MRCT.
A later MRCT website listed Mark Barnes and Barbara Bierer as its founders. Barnes is a lawyer variously linked Ropes and Gray, Harvard, and Pfizer. Bierer is a professor of medicine (haematology) at Brigham and Women’s hospital.
A year after signing the above in 2018 Agreement Pfizer produced a massive document stating in minute detail how their organisation is controlled by them. It’s a VERY long document .
The Orange Guide .
Living our value of Equity requires that we act with integrity. One way we demonstrate our
commitment to integrity is by complying with laws and the rules governing our business.
Compliance with these laws builds trust with patients, Healthcare Professionals, institutions,
purchasers, and the government. It is also critical to achieving our purpose of breakthroughs
that change patients’ lives.
Copyright © 2019 Pfizer Inc. All rights reserved
This Orange Guide has been updated in 2021
(PS the MHRA also published Orange Guides – what’s with the colour orange – should be brown surely)
FDA vaccine adviser warns healthy young people should NOT get new COVID booster: Says it’s ‘unfair to make them take a risk’ after data suggested shot was not as effective as first batch
Dr. Paul Offit, a member of the FDA’s Vaccine Advisory Committee, said he’s not fully sold on the benefits of a third shot outweighing the harm
A newly developed dose, called a bivalent vaccine, is a cocktail of the original coronavirus strain combined with parts of the omicron BA.4 and BA.5 subvariants
The CDC has reported that the old vaccine has side-effects like myocarditis, an – inflammation of the heart muscle and pericarditis, an inflammation of the heart’s outer lining
So far, the only tests on the new shots have been done on lab mice
By JANON FISHER FOR DAILYMAIL.COM
PUBLISHED: 05:18, 25 September 2022 | UPDATED: 11:38, 25 September 2022
https://www.dailymail.co.uk/news/article-11247181/FDA-vaccine-adviser-says-healthy-young-people-SHOULDNT-COVID-booster.html
A top vaccine expert and pediatric doctor is cautioning parents of healthy young people to hold off getting the new COVID booster shot, saying it can carry risks and its efficacy hasn’t yet been proven.
Dr. Paul Offit, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia and a member of the Food and Drug Administration’s Vaccine Advisory Committee, said he’s not fully sold on the benefits of a third shot outweighing the harm.
‘Who really benefits from another dose?’ Offit said on CNN.
‘Right now they’re saying we should trust mouse data,’ he said, ‘and I don’t think that should ever be true.’
From Annie’s comment above Dr Aseem Malhotra
@DrAseemMalhotra
‘….. The paper below highlights just how corrupted the system has become by psychopathic entities’
Corruption Applies to the whole of so called ‘health care’
So yet another death yet another cover up yet another stringing out the investigation by so called care quality commission It never ends
Mental health hospital is rated inadequate two years after patient’s death
BMJ 2022; 378 doi: https://doi.org/10.1136/bmj.o2325 (Published 26 September 2022)
Woodbourne Priory Hospital in Birmingham has been rated inadequate by the Care Quality Commission after a surprise inspection in May.1
The inspection was carried out in response to a coroner’s publication of a document on the prevention of future deaths after a hearing into the death of Matthew Caseby, who was using services at Woodbourne in September 2020.
The CQC’s inspection report, published on 22 September, saw the hospital’s rating for safety decline from “required improvement” to “inadequate,” while the rating for whether it was “well led” fell from “good” to “inadequate.” Inspectors found the …blah blah – we can guess the rest of it.
The nastiness and abuse reflects the attitude on all levels by some who work and control people in institutions towards people regarded as inferior human beings. The college expects congratulations for contributing to this exhibition? Interesting they call it an exhibition. It won;t change a thing for people incarcerated or caught up in the hell of psychiatry
Exhibition: “We Are Not Alone:” Legacies of Eugenics
Launching on the 22nd of September 2022, we are hosting an exhibition called “We Are Not Alone”: Legacies of Eugenics.
The exhibition was created by Professor Marius Turda (who is not a clinician)and was first hosted at the Wiener Library in London in September 2021. Since then it has travelled to Romania, Poland and Sweden and in April 2023 will be visiting Harvard University. Also beginning with the end of September 2022, UCL’s Institute of Education will also host sections of the exhibition dealing directly with education.
The History of Psychiatry Special Interest Group( whose members are mainly retired) has worked with Professor Turda to add a panel discussing the RCPsych and its members’ involvement with eugenics, in the hope that by accepting and exploring our history we can better challenge systemic inequality and prejudice in healthcare today. (Self delusional
tosh they can hardly honestly believe themselves but it sounds good)
“It won;t change a thing for people incarcerated or caught up in the hell of psychiatry”
Yes Susanne. And that area is really up market, half a mile outside the centre. They don’t want the ‘loonies’ getting out and the hell of psychiatry ostensible to the rich upper middle class who live there.
“of concern highlighted for action by the Priory Hospital. These were record keeping, record keeping quality, risk assessments, serious incidents and the security of the courtyard fences”
“The coroner had highlighted five areas of concern impacting on the safe care of patients and contributory to the death of a patient in September 2020.”
So they listened to the coroner, they don’t listen to the patients nor really care about the patients that’s for sure. They care about containing a massive many decades long scandal in all of psychiatry going right to eugenics.
https://api.cqc.org.uk/public/v1/reports/a3a2b217-f789-41f2-bc2f-38f03fa70d39?20220922070310
I’ve never read the word AKATHISIA in any CQC document nor Heath Trusts public documents. Until they face up to AKATHISIA caused by doctors forcing antipsychotics and what it really is, it will continue to get worse and the CQC will cover up, if it can’t cover up – in this case it seems to be the area – they will shut it down.
HART
@hartgroup_org
Health Advisory & Recovery Team. UK doctors, scientists, economists, psychologists and academics. Sharing concerns and widening the debate on COVID-19 policy.
Dr Ros Jones
@MaidmentRos
and
@ClareCraigPath
on
@GBNEWS
with
@MarkSteynOnline
They are urgently asking the government to pause the vaccine campaign in healthy children. There are worrying signals in the data which need to be investigated.
https://twitter.com/hartgroup_org/status/1574487928843091968
HART member Dr Liz Evans and
@ukmfa1
founder explains the importance of weighing up risks vs benefits and also the importance of medical ethics
@trussliz
@DHSCgovuk
we urge you to reconsider your position based on current evidence and worrying signals from international data
https://twitter.com/hartgroup_org/status/1574489794670903296
LIVE
Press Conference:
Dr. Aseem Malhotra’s New Peer-Reviewed Papers Lead Calls for Immediate and “Complete Suspension” of All Covid-19 Vaccines
Consultant Cardiologist Dr Aseem Malhotra will present new evidence relating to the safety and efficacy of mRNA Covid-19 vaccines. This will include analyses that answers the question ‘is there a link between these new types of vaccines and an increase in cardiac events?’
Broadcast started 9/27/22 10:30am (ending 9/27/22 2:00pm)
https://worldcouncilforhealth.org/newsroom/
FAB U LOUS…
It has become clear to me, that medical care is about enriching corporate medicine. Whether one dies or not is not relevant to those who practice it. There is control of the narrative, by the corporations, and even though people are dropping dead left and right, you are the crazy one, for refusing care. I expect that at some point there will be punishment for resisters. A punishment that will become harsher, mote violent as time goes on.
A good number of people in the world apparently will do as they are told, even unto their deaths. But there are others, who will resist, in this life and death struggle. Deaths will have to be covered up, and blamed on other diseases. For obvious reasons. I expect that at some point it will be prudent to wear body Armor, to protect oneself from being injected, by those who want to take everyone else with them.
There are remedies, I suggest stocking up now, the future doesn’t look bright. I myself have avoided medicine for many decades, and I am healthier for it. So many medications have been withdrawn that turned out to be mass killers, despite lengthy approval processes, it just was not worth the risk. Sunshine is free.