
Some months ago I was asked by Paul Rosenfeld, who directs psychiatric training at Mount Sinai Hospital Morningside West, to give a lecture. This took place on January 18. There was a Q and A afterwards. The talk was recorded and the video is here – Relationship Based Medicine.
There will be two related talks, which will also feature in posts soon. Relationship Based Medicine was delivered to clinicians. The next is an open public lecture, which will take place in Lethbridge Alberta today – Thursday January 26, and is called Beware Doctors Bearing Gifts. The final one will be in a few weeks time to a mixed group of lawyers and doctors. There are some overlaps but each talk brings different issues into play.
As per the YouTube and Podcasts message, it would be good if people could subscribed to the YouTube channel – subscribe does not mean hand over money.
The slides and text of Relationship Based Medicine – the original title for which was Standardizing Psychiatry and its Clinicians (Slide 1) – are below.
Slide 2: The date on this suicide note is October 2004. Concerned that FDA planned to put a Black Box Warning on antidepressants for minors APA state they believe ADs save lives.
Saying suicide note or as I will say later the Art of Medicine involves bringing good out of the Use of a Poison might make you think I’m anti-drugs or the medical model. I use medicines and ECT rather than any other form of treatment and believe in the medical model. But the art of using these treatments also means knowing when not to use them.
Slide 3: APA should have said they believe Clinicians save lives. The magic should lie in you not in pills.
But the processes of standardization affecting all of health festoon antidepressants with numbers. These look like science but aren’t. You make or should make judgments and choices, which cannot be standardized. These do not look like science but are.
In addition, making judgments is a moral enterprise – simply using antidepressants is amoral.
Slide 4: The crisis bothering APA began 15 years earlier with this paper, in which 3 clinicians claimed fluoxetine caused 6 people to become suicidal. The causal claim was based on analyzing the cases closely and followed traditional clinical approaches for determining causality. There were about 10 other groups reported similar findings.
Slide 5 I contributed to this debate, outlining the cases of 2 men, challenged with an SSRI, then dechallenged and rechallenged. There was no other way to explain what happened except that fluoxetine had caused it.
Slide 6 Almost the same week this Lilly article came out in the BMJ claiming an analysis of their clinical trials showed no evidence fluoxetine made people suicidal. The cases reported were sad but anecdotal and the plural of anecdotes is not data. Depression was the problem not fluoxetine. Clinical trials are the science of cause and effect.
This paper created Evidence Based Medicine and the question to doctors, the public, the media, politicians was – are you going to believe the science or the anecdotes?
In fact, the original phrase is the plural of anecdotes is data – otherwise Google wouldn’t work.
The idea the disease was responsible for suicide attempts and suicides in healthy volunteers or patients in trials with anxiety or OCD was hard to believe but some experts claimed just that.
Finally, what I hope to show you is the Teicher et al paper was Evident Based Medicine whereas Lilly’s Evidence Based Medicine is artefactual.
Slide 7: The Art and Science of Medicine are often contrasted. Those in the soft human sciences contrast their rich qualitative context with the experimental context, where hard scientists eliminate confounders with RCTs. This pre-Risperdal Janssen Advert assumes you will figure that paying heed to the soft side of things is a touch twentieth century.
My argument is the Science of Medicine lies in making hard judgement calls. A made by algorithms medicine may make your life easier but will doom Us. By Us I mean the chance of a community. Following the Artefact alienates you from Us.
Slide 8: The origin of science story usually starts in 1660 when The Royal Society came into being. This established the ground rules of Science.
Science would deal with philosophy or theology. It would only deal with matters that could be Settled by Data. The focus since has been on the Data, but participants were called on to make judgement calls about the data in front of them. If you were Xtian, Hindu, Jew, Muslim, or Atheist, you left these badges at the door and had to explain what you saw without appealing to a Book or Authority.
You could run the experiment again, adapt the apparatus, check it out for tricks – what you couldn’t do was refuse to come to a Verdict about the data.
Slide 9: This account of our history overlooks an equally important event 44 years earlier, when Walter Raleigh had his head chopped off – supposedly for being too close to those pesky Europeans. Raleigh was convicted on the basis of things said about him by people who did not come into court to be cross-examined.
Legal systems worldwide recognized the injustice of this and introduced Rules of Evidence. Hearsay could not be used as evidence. The only evidence that can be used in court is material that can be put in front of jurors who can see the people or material being examined and cross-examined. The process of forcing 12 people with very different biases to come to a Verdict about what is in front of them is the essence of science.
Verdicts and diagnoses are provisional – the view that best fits the current facts. If the facts change, we reserve the right to change our mind. This might appear to contrast with the objectivity of science, but scientific views are similarly provisional. Scientists attempt to overturn verdicts with new data.
If I gave fluoxetine to Paul 30 years ago and he came back suicidal. I could examine and cross-examine him, run lab tests and scans, raise the dose, stop the drug, add an antidote, check with colleagues has anyone else seen anything like this or can they explain it in any other way.
If Paul and I conclude fluoxetine has made him suicidal and report this to FDA, the first thing FDA does is to remove his name. No-one can now examine or cross-examine him and come to a scientific view about whether there is a link or not. His injury has been made Hearsay.
If you are later injured in the same way and see 1000s of reports of suicidality on SSRIs on FDA’s adverse event reporting system, you cannot bring this evidence into court because no-one can be brought into court. It’s Hearsay not evidence.
Company assays are all Hearsay – they should not be let into Court as evidence. Names count. Paul in this case is the data and accessing the data means accessing him.
If Paul and I report his case in a journal as a Case Report, with our names on it, either of us can be brought into Court. Case reports are the best evidence we currently have in clinical psychiatry – the only material that reaches standards of evidence.
Slide 10: Here 20 years after he created the first RCT, is Tony Hill saying RCTs can be helpful for assessing one of the 100 things a drug does – and this might be useful for treatment purposes.
If RCTs evaluate only one of a 100 things a drug does, by definition they are not a good way to evaluate a drug. Hill was clear that clinical judgement should not be replaced by an algorithm.
All RCTs generate ignorance, but they can be useful provided we remember that some can also generate useful information – we can bring good out of this artefact.
Slide 11: This 1960 placebo controlled RCT makes Hill’s point well – thalidomide has therapeutic efficacy as a sleeping pill but the trial shows nothing of the SSRI-like sexual dysfunction, suicidality, agitation, nausea and peripheral neuropathy thalidomide causes. Louis Lasagna was the person two years later responsible for incorporating RCTs in the 1962 Food and Drugs Act amendments.
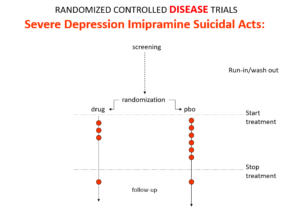
Slide 12: Some people say RCTs can demonstrate cause and effect in a way that other study designs can’t. Imipramine was the first antidepressant. It and other tricyclic antidepressants are stronger than SSRIs and SNRIs. They beat SSRIs in RCTs. They can treat melancholia – SSRIs can’t. Melancholia comes with a high risk of suicide.
Imipramine was launched in 1958. A year later at a meeting in England, Danish psychiatrists made it clear that while it was a wonderful treatment it made some people suicidal.
Let’s do a thought RCT of imipramine versus placebo in melancholia. Even though it can cause suicide, we would expect it to reduce the number of suicides in a trial like this because it treats the condition. This RCT would be great evidence antidepressants do not cause suicide.
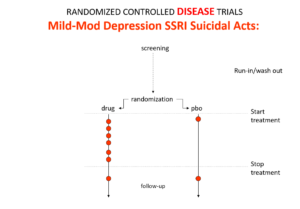
Slide 13: Here is the data on the trials in mild depression that brought the SSRIs and SNRIs on the market – you see a doubling of suicidal events compared to placebo. Companies resorted to all sorts of illegal maneuvers to hide this risk.
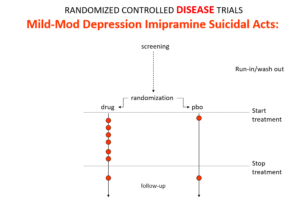
Slide 14: This is what the data for imipramine look like in the same mild depressions. Now it seems that it too causes suicides. So RCTs tell us nothing about cause and effect – they can give us diametrically opposite answers. This is because these aren’t drug trials. They are Treatment Trials and in any clinical Trial, the condition confounds the effects of the drugs – and these confounders hide drug effects.
People evaluating drugs in traditional clinical trials, before RCTs, knew this. People doing RCTs don’t. When a patient becomes suicidal in a trial you have to use your judgement to work out what is happened but in RCTs clinicians are not supposed to use their judgment.
This is not just the case for depression – it’s true in every clinical situation where drugs and conditions cause superficially similar effects – diabetes and glitazones both cause heart failure, osteoporosis and bisphosphonates both cause fractures
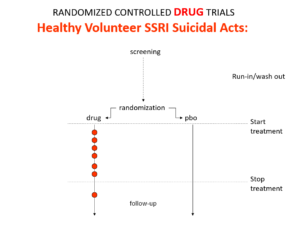
Slide 15: Here is what a drug trial looks like. Companies ran these studies in the 1980s and found that SSRIs make healthy volunteers suicidal, caused dependence and sexual dysfunction but we heard nothing about these problems when the drugs launched. These Drug Trials enabled companies to engineer their Treatment Trials to hide these problems.
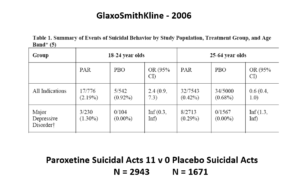
Slide 16: Other people say randomization controls unknown confounders. This slide shows some data straight from a 2006 GlaxoSmithKline paper. GSK’s SSRI paroxetine was in trouble – the RCTs data for Major Depressive Disorder seem to show paroxetine causes suicidal events. The real data I think are worse that GSK admit to here.
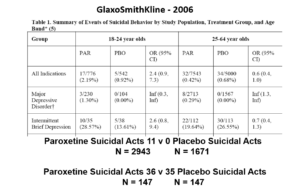
Slide 17: But never fear RCTs come to the rescue. GSK also did trials in people with Intermittent Brief Depressive Disorders – IBDD. These are borderline personality disorder to most people – patients who have suicidal events much more often than anyone else. But these patients can meet criteria for depression and could be entered into Depression RCTs.
GSK did two trials of paroxetine in this group – the data didn’t look all that good. Here are IBDD data from the two GSK trials.
Slide 18: When you add the IBDD data to the MDD data – all of a sudden paroxetine doesn’t cause suicidal events, it protects against them. We could add 12 more events to the paroxetine arm and still get the same magical outcome and 40 events before the company would have to concede their drug caused people to be suicidal.
Something like this is going to happen in every treatment trial where the patients entered are heterogenous – back pain, breast cancer, diabetes, hypertension, osteoporosis, parkinson’s disease. We can use an effect a drug causes to hide an effect a drug causes. RCTs are not a good way to work out what is going on.
Slide 19: Let’s look deeper into RCTs. The first rating scale for behavior was the Hamilton Rating Scale for Depression. Here is Max Hamilton saying:
It may be that we are witnessing a change as revolutionary as was the introduction of standardization and mass production in manufacture. Both have their positive and negative sides
He saw the scale as a checklist of things to ask about in an interview – a mixed blessing.
Slide 20: If you cleave to the checklist, you will do standardized but possibly disastrous interviews. For instance, on the scale, there is a suicide item. Suicidality can stem from the illness or the drug. This needs a judgement call – if caused by the drug you should rate the person as Zero – if caused by the illness you might rate 3 or 4. Ditto for sex, for sleep. If you just check yes for suicidality, the default is to the illness.
Checklists like these however are now viewed as scientific instruments. They look better to hospital managers than David Healy asking you about the football last night or your family.
For drug companies, rating scales ensure you do an interview that produces figures which are the most seductive way to get people on their drug. Your interview will help you to help your patient to live the life Pfizer want him to live. Do that and you are no longer practicing medicine.
Slide 21: Pfizer know all this very well. Here 15 years ago is an advert for a Pfizer symposium at an APA meeting. Skills are second millennium. Scales are the new go-to.
Slide 22: Three months ago, the New York Times and New York Post carried the same story – US teens were on up 10 psychotropic drugs at a time and the outcomes were not good. There was no good explanation for how this was happening but its clear that one pathway is clearly rating scales. I’m sure all of you have seen the patient who was put on an antidepressant and becomes suicidal or agitated and the response is you’re bipolar – and lets begin you on a mood stabilizer anticonvulsant or antipsychotic.
So, as we are creeping up from 1 or 2 drugs, the person gets put on an antipsychotic which will cause everyone to lose focus – that’s what we want them to do – and they mention on the next visit they have lost focus a bit – so we give an ADHD rating scale and all of a sudden they have ADHD and we add a stimulant. This substituting scales for judgements is like putting someone on a weighing scales – handing them 2 25 kg dumbbells to hold and saying hey you’re overweight – we need to give you a stimulant for weight loss.
Slide 23: This is true for any measure – peak flow rates, bone densities, blood pressure or lipids, or sugar. You may need to do something about some figures, but the goal is to help people to live the life they want to live – not get them to live the life Pfizer want them to live.
Up until the 1980s, people brought their problems to us – seeking help to live the lives they wanted to live. After that we began to give them problems with lipids and other risks and the amount of medicines consumed rose dramatically. We began treating numbers rather than people.
Just after weighing scales were introduced, we got the first descriptions of anorexia nervosa. In the 1920s, weighing scales in drug stores had norms for ideal weight attached to them and eating disorders mushroomed. They migrated into our homes in the 1960s and eating disorders became epidemic.
Measurements risk becoming an illness. It’s great if we can remain on top of the measurements from one fraction of our lives – like using a stopwatch to break our personal record for the mile – but if we can’t remain on top of it we create a neurosis.
Can we leave bone densities thin, peak flow rates low, lipid levels high? Yes, we can. We think post-mortems reveal what we died from – they more often reveal what we can live with. Is it riskier to live with raised lipids or to ingest ego alien chemicals and ego alien ideas every day?
Slide 24
There is an extra element to the equation. The Service industries emerged in the 1950s. Through to 1990 or so, no-one viewed health as a service industry – doctors were professionals who exercised judgement the way a Judge might. Service industries have managers. When health became a service industry, we got managers. Before that our exercise of clinical discretion was a jewel in the crown of Care, but it is a problem for those who manage services and our contracts in many places now stipulate that we adhere to Guidelines that in the case of drugs are deeply problematic. We used to manage the risks patients had to help them live the life they wanted to live – we now manage the risks they pose to the service we work for.
Slide 25: Managers manage what they can measure. The figures have a sheen of scientific gold
But in a modern version of the Midas Story this Gold is killing Care
Slide 26: The crisis that concerned APA started good and proper in 2001 with this article which has an authorship line to die for, in the Journal with the highest impact factor in child psychiatry reporting Study 329 a trial of paroxetine in pediatric depression claiming it works wonderfully well and is safe.
Slide 27: Three years earlier, in 1998, GlaxoSmithKline were mulling over this document which says Study 329 was negative for efficacy and safety. What were they going to do? They were going to pick out the good bits of the data and publish them. The good bits formed the Keller et al 2001 paper.
This 1998 document led New York’s Attorney General to file a fraud action against GSK. As part of the resolution of this, GSK agreed to make public the their paroxetine trial data public. A decade later, GSK resolved a Dept of Justice action, which also involved Study 329, for $3 Billion dollars.
Slide 28: After the $3 Billion settlement, a team of us got together to Restore Study 329. By accident we got Appendix H which contains the Clinical Report Forms – CRFs. Over 80,000 pages. Analyzing this does not require much expertise. If you could print it off and spread the bits out and let some College students loose on it, they’d be able to tell you what this Study really found.
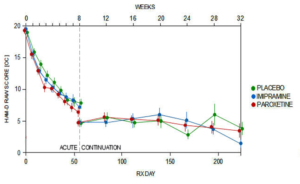
Slide 29: The 8-week acute phase of the study showed no difference between paroxetine, imipramine and placebo. Ditto for the never published 6 month continuation phase.
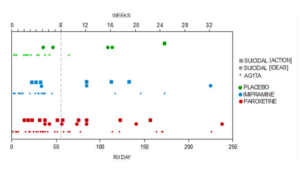
Slide 30: Paroxetine had over 500 side effects. The Keller paper admitted to half of these. Keller noted 6 emotionally labile events in the trial, some of which might have been suicidality, 4 on paroxetine. But nearly a quarter of the children in the trial had a behavioral event on paroxetine mostly suicidality.
Slide 31: Suicide is not what I want to focus on. It’s the ability of company assays – they are not trials – to hide adverse events. Coding is top of this list – this is the first act of authorship but no one pays any heed to it. Let’s say a man on a drug gets agitated, pours petrol on himself and sets fire to it intending to kill himself but only dies from his burns 5 days later. You can code him as death by burns or suicide. If the coder opts for death by burns, the data will look very different. This is a real example – just as emotional lability was.
About 15% of events disappeared because they were not transcribed to the final datasheets.
Let me take you to grouping. Rather than list out all events, they are usually grouped under say Cardiac or Gut. In the case of behavioral events, these could be put under Neurological or Psychiatric Events. GSK grouped all behavioral events under Neurological. This groups emotional lability with headaches and dizziness, which are very common and grouped this way behavior problems disappear. Put behavioral events into a Psychiatry group and all of a sudden you see the problem.
Slide 32: The Restoring Study 329 article had 7 reviewers and 7 rounds of reviews. It took over a year to get it published. We were having problems with the BMJ research editor, Elizabeth Loder, who blew a fuse every time we tried to move behavioral events out of the headache group and into a Psychiatry group.
Turns out Dr Loder is a neurologist and Headache expert. She has worked on GSK’s migraine and headache meds. She also has a husband, who is a lawyer and worked for the law firm that defended GSK in their $3 billion Dept of Justice case. Maybe no link but when we pointed this out, the article was accepted.
Slide 33: The full story of Study 329, with the document you have seen, and one you will see soon, and many others are all on this Study329.org website.
Slide 34: The even fuller story can be found in Children of the Cure.
Slide 35: The crisis about these drugs blew up in 2004. In 2002 FDA had sent GSK an approvable letter for paroxetine in pediatric depression. This says GSK have told them Study 329 is negative. FDA agree its negative – in fact all 3 trials are negative. FDA will still approve Paxil for kids. FDA also agree with GSK’s suggestion it would be better not to mention the negative trials in the label of the drug. Why would FDA agree to this?
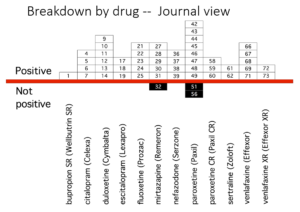
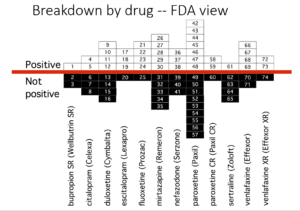
Slide 36: This slide from Erick Turner’s famous 2008 article shows published adult ‘trials’ on the antidepressants we know and use. Almost all indicate the drugs work well and are safe.
Slide 37: Another slide shows the trials as FDA viewed them. Many trials published as positive were negative to add to the unpublished negative trials. Meta-analyses of these drugs now pitch sertraline as the drug to take – but this slide suggests it’s one you should avoid, which is consistent with the great difficulties sertraline had getting approved by regulators.
Slide 38: Companies run assays to get on the market, not trials to inform clinical practice. From the 1980s companies took the running of assays out of the hands of people like Paul or me and gave it to Contract Research Organizations (CROs) now a $40 billion business. Once Paul and I had the data in our filing cabinets, now CROs have it and no clinician sees it. FDA don’t see it either.
The writing was given to Medical Writing companies. The true author of Study 329 is not listed among the 24 or so authors you saw on that paper.
What does that mean? Here you see the PTSD page of a document listing Zoloft articles in progress. These aim at capturing markets not at informing you how to use Zoloft safely. You see on the right two articles are complete and will be sent to the very best journals – on the left you see TBD – to be determined – when Pfizer decide which names would sell most Zoloft.
Pfizer did 4 Zoloft PTSD trials. All 4 were negative. But 2 had a minimal benefit for women – these good bits plucked out are what’s being published. Tough on the troops coming home.
Why do FDA say nothing about these negative trials published as positive? Well if FDA said trials are negative – the companies might get sued for fraud – as happened for Study 329.
FDA will tell you – it is not their job to police the medical literature. What is FDA’s job?
Slide 39: Some years ago, Britain joined the EU and ran into trouble. They were told that Cadbury’s chocolate could not be called chocolate. It didn’t have the right quota of cocoa solids. British consternation over chocolate led to Brexit some decades later.
FDA regulate Food and Drugs. Faced with butter or chocolate or drugs, companies must meet an assay standard – so much cocoa or so many points on a Depression rating scale in 2 trials. Meet that and FDA let you use the words chocolate, butter, or antidepressant. It’s not their job to decide if this is good butter, or if chocolate is good for you, or police the drugs literature.
FDA even said at the Zoloft depression approval hearing – if a company has two positive assays, even if 98 others are negative, sticking strictly to the wording of regulations they could let Pfizer call this an antidepressant.
Slide 40: Where does this leave us? Back in 2016, you have BMJ featuring Jeremy Hunt, then the UK Minister of Health who said:
Children’s mental health is possibly the biggest single area of weakness in NHS provision at the moment.
This is hardly surprising. In 2004 the entire pediatric psychotropic meds literature was company written and constitutes the greatest divide between the published claims and what the data show in any branch of science that I know of.
In particular the literature and regulators and politicians say Prozac is fine for children. Well according to FDA reviews, these trials were also negative. The data are just like Study 329, but it got approved before the crisis blew up and regulators didn’t want to – you’ll have to ask them.
Slide 41: When I was a teen it was accepted we went half-crazy as we adapted to a crazy world. It was thought important for us to go through this, as Homer tells Bart.
Now Homer would be encouraged to give Bart a rating scale and based on the data take him to a doctor. Greta Thunberg and her generation, who are doing great things to stop us pumping chemicals into the environment, are pumping more chemicals into their inner environment than any prior generation.
Slide 42: Here is FDA again approving products.
Slide 43: Let’s say you break a limb and go to an ER where they are doing an RCT of plaster-casts – randomly applying a cast to one of our four limbs – this trial will show randomly applied casts beat placebo, but it would not be intelligent to practice medicine this way. The slide also reminds you you have 4 limbs.
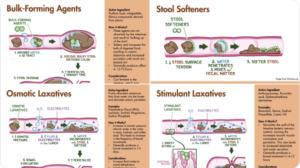
Slide 44: There are also 4 therapeutic principles for managing constipation. There is no point having someone on all 4 and it may be dangerous. One is probably key. The clinical skill lies in knowing what the right approach for your patient is. The risk is thinking the person has bad constipation because the first 3 didn’t work when this shows out lack of diagnostic skill.
Slide 45: The same applies to Antihypertensives – there are 4 therapeutic principles and to hypoglycemics and most other drug groups in medicine.
Slide 46: The same is true of antidepressants. An antidepressant label gets doctors and the public thinking they all do the same thing. But SSRIs have a completely different effect to noradrenaline reuptake inhibitors, or drugs like trimipramine or mirtazapine – and anticholinergic drugs, which are euphoriant, are more effective for melancholia than SSRIs. There is no skill in giving an antidepressant. The skill lies in knowing what therapeutic principle this person needs.
Companies have made more than $100 billion from these drugs and we still don’t know what to give the person in front of us – there is a 50-50 chance they’ll get put on the right drug for them. I wonder if thirty years ago companies thought they would get such an easy ride from us.
Slide 47 A lady I saw years ago had dropped out of High School early and had no background in healthcare. She was put on an SSRI. She ended up convicted of drunk and disorderly behaviour, crashed her car, was jailed, lost her job. She figured it was the SSRI she had been put on. Her doctor and AA told her this line of thinking proved she was an alcoholic.
She was right and they were wrong. It took years but she went on the internet and ended up being able to tell me things about the serotonin system and what drug she should be put on instead of an SSRI. The only people who agreed with her were the pharmaceutical industry who were busy working on just what she found were possible treatments for alcoholism.
What I got from this is that Motivation is worth more than Expertise. Rather than seeing 100 heart-sink patients in my waiting room, heart-sink because they aren’t responding the way a ghostwritten Literature tells me they should – I could see 100 free researchers who could help me discover things I never knew about. The job could be fun.
Another patient had OCD. We tried the full range of standard treatments but nothing helped. Starting with his own experience, he found restarting smoking helped. He then researched trials of the benefits of nicotine for OCD and found several showing effects comparable to SSRIs – something that would never have occurred to me – or I guess you.
This is clearly bringing good out of the use of a poison. Staying on treatment every day for the rest of your life means balancing risks and benefits
The image here shows another problem – Post SSRI Sexual Dysfunction – PSSD. This happens not infrequently. The sexual dysfunction that happens pretty well everyone on an SSRI gets worse on stopping and can last for decades.
The condition leads to suicide and the fact there is no research happening on it – although there is a Nobel Prize to be won for finding the answer – also leads to suicide. The people doing or funding the research are those affected – so there is hope for an answer.
But here’s another point. Regulators these days claim they approve drugs on the basis of a favorable benefit-risk ratio. This assumes a mood benefit is the commonest thing an antidepressant does when it’s quite rare – and when in fact close to 100% of people get genital numbing within 30 minutes of a first tablet of an SSRI and most people have some sexual dysfunction.
Letting a company call a drug an antidepressant is one thing but claiming a favorable Benefit-Risk ratio is something regulators and companies should not be able to do – it is down to you and your patient to decide if the Benefit for this person is worth the Risks they run.
Slide 48: At present the public and politicians and many doctors etc see FDA as super scientists. They are not. If anyone with medical training joins FDA, it’s because they don’t like meeting people. But as one ex-FDA senior official put it to me – a Delegated Narcissism effect comes into play. Most people want there to be a Father Figure or God somewhere in the background to stabilize the system we are operating in.
Slide 49: This image better characterizes the role of FDA today. Regulators permit the use of the word antidepressant. Back in 1962 when these regulations were put in place, it was clearly understood that doctors were supposed to keep us safe.
Slide 50: Doctors are the people who are supposed to exercise judgement. It’s now a scandal if you exercise Judgement. The magic of medicine used to lie in bringing good out of the use of a poison – based on your judgment call. Because you no longer exercise judgement drugs we once knew were poisons have become sacraments – a sacrament is something that can only benefit – it cannot harm.
Specialists have become partialists. Boxologists. Unable to cope if a condition includes something outside our area of expertise – but it is almost the definition of toxicity that the problem affects lots of or most bodily systems.
Slide 51: I have used the word poison again. The origin lies with Paracelus who said everything is a poison, it is the dose that makes it possible not to poison someone. We used to have poison symbols on meds but now we have serious proposals to stamp pills like Diclectin or SSRIs (which cause birth defects) with a soothing image of a pregnant women.
An intriguing feature of the Paracelsus saying is that Gift is the German for poison. Beware Doctors Bearing Gifts.
Slide 52: I used to compare doctors to pilots – both have adverse event reporting systems. When pilots report, safety systems pay heed because they know she won’t fly if they don’t because if you die, she dies. Doctors in contrast fly on – if you die they can blame the disease.
The third photo features Jane Frazer the CEO of Citibank. She moved into a senior position at the time of the financial crash. She looks like she did her job well because she has been promoted ever since so she may not be guilty of the moral hazard bankers were guilty in the crash. Their job was outsourcing risk in the knowledge that if things crashed, the bank would repossess your home and bankers still get bonuses while you lost everything.
Doctors outsource risk as well – and this is morally hazardous. Our job used to be to take risks on behalf of and with patients in order to help them. It is now more often a case of managing risks to an organization we have to put before patients.
Slide 53: This image from 2004 shows that many people back then could see that doctor’s days were numbered. If the drugs work very well and are free of hazards, then doctors are expensive prescribers – nurses will do the same job and cost less.
If we are going to have a place in clinical care, we need to remain salty – we need the system to know these drugs are risky and someone needs to be paid more to handle risky chemicals and ensure that good is brought out of their use. But bringing good out of the use of a poison does not compute for managers, or insurers, or politicians, or the public. Have we left it too late?
Slide 54: Back in the last millennium, the Institute of Medicine produced a report on the increase in people dying from medical errors. They wanted us to put safety systems in place.
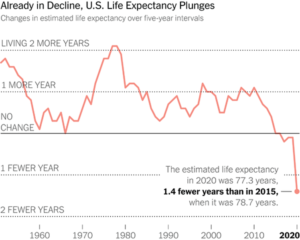
Slide 55: Two decades later, things are much worse. Here is a recent New York Times image of Life Expectancy in the US, which has been falling you’ll note well before COVID. My view is that this is most likely linked to polypharmacy. The UK has similar falling Life Expectancy data – again pre-COVID
Slide 56: Drugs are like guns – they can be very helpful in certain situations. But we sense that letting them multiply up and leak into every situation is a recipe for problems. Drugs and Guns are techniques. Guns create an arms race.
Medicine has been driven forward by a military need also – losing more men to disease than to combat was the norm until recently. The military need was to be able to keep men able to fight.
Guns and drugs are amoral. Whether they enhance or diminish us is down to us. If we stop thinking about what we are doing when we use them, we are highly likely to be diminished.
RCAs are also a technique. They create the information that should transform chemicals into medicines. At the moment, the drugs race is not about creating better chemicals – it’s about creating more effective propaganda. By definition this diminishes us.
Slide 57: The arms race brings out an important message. Efficacy has its limits. We can make weapons more and more effective to the point that they can’t be used. The same holds true for drugs. If you are on more than 3 medicines, effectiveness diminishes. To get the most effectiveness you need to be on 3 or less. As of 2016, over 40% of over 45 were on 3 or more drugs every day of the year – this figure includes the people who never come to see doctors. Over 40% of over 65s are on 5 or more drugs every day of the week.
We know that life expectancy is falling. We know that reducing medication burdens can increase life expectancy, reduce hospitalizations, and improve quality of life.
Paring our rapidly escalating medication burdens back to 3 or less should mean taking the values of the Your patients also face a Tiananmen moment with you as the tank driver. We get nasty when patients bring us adverse events because we are scared regulators, guideline makers, colleagues, politicians and the media will be nasty to us. Even Greta Thunberg’s generation will mobilize against us.
Slide 58: Reducing a medication burden is not easy – as this image from the movie The Hurt Locker attempts to portray. For many people, few of the drugs they get from us in mental health are likely to reach the Top 3 or 5 they might need to be on at some point to stay alive. Many of the drugs we give them are ones it can be terribly difficult to get off should they need to reduce their medication burden when thinking about adding other necessary drugs.
There will never be any RCTs to help us out here – the best evidence will lie in good case reports and our ability to listen to and look at people. If we don’t listen to and look at we are not doing science.
Slide 59: If you try to remain salty – you risk having a Tiananmen Square moment.
If you try to modestly reduce medication burdens or recognize that in some cases a treatment might have become a problem – as in someone put on an SSRI in their teens and 10 years later it’s pooping out for which we have no treatment – you are likely to be told that what you are doing would be great in private practice where you could let people know what you do and they can choose it, but here the expectation is people want more diagnoses and drugs.
To get treatments to save our lives we used to have to be wealthy. To reduce medication burdens now in order to save our lives is now a Privilege of Wealth.
Slide 60: Canada now leads the world in MAiD – Medical Assistance in Dying. Doctors can be struck off if they don’t make an effective referral for this new service – patients get much more attention and input if they apply for this new service. But if you want the service of having less services well the Canadian system does not want to know you.
Slide 60: This lady is in our waiting room. You might see her as one of the people we call heartsink patients – we stick to the guidelines but she doesn’t get better.
She comes from an Arthurian Legend. Arthur has been out-fought by a Black Knight who spares his life if he can answer a riddle – What do Women Most Desire. He has a year to find the answer. His court hunt desperately for it. En route to meet the Black Knight, Arthur and his troop meet this woman who tells him that she can give him the answer to the riddle but one of his knights must become her husband. Gawain offers himself. Arthur answers the riddle and a furious Black Knight lets him go.
Slide 61: Gawain gets married. Everyone at the Court is unhappy for him.
Slide 62: Gawain can’t bear to look at her. She takes control and he finds a most beautiful lady, who asks – do you want me to look like this by night with you or by day in court. He says – whatever you want. Which turns out to be the right answer.
The answer to both riddles is she, like us, wants to control her own life. Help her live the life she wants to live, and she becomes a most interesting and, in that sense, attractive person. Perhaps a disease needs treating – but she doesn’t want you to tell her how to live a life, or want her negative emotions eliminated with a pill. She may be doing better at living life than you or I.
A relationship based medicine is the only scientific form of clinical practice. Rather than 100 heartsink patients it offers us 100 free research assistants, whose motivation often counts for more than expertise.
Both science and morality depend on collaboration. The more you collaborate, the more interesting the people you work with become which leads you to appreciate them more and them you. This virtuous circle creates an Us that leaves us all better placed to live the life we want to live.
Evidence based medicine creates a False We – it has you relating to a non-existent average person rather than the person in front of you taking a drug – who is the apparatus in which the experiment is taking place and your job is to explain what you see happening in front of you – and you are not supposed to bring books and authorities into the room.

















































A year and a half ago our Commander-in-Chief told us “You won’t get covid if you have these vaccinations.” They’ve stopped pretending that. They’ve also quietly dropped the selling point that the mRNA shots prevent transmission, and now are reduced to arguing that the shot prevents severe illness and death due to covid.
And how do we know this? Not from randomized controlled trials, which were shut down after something like seven percent of participants made it to the six-month mark. No, this comes from data from the dashboards of public health agencies, who don’t tell you they are not counting events in the “partially vaccinated,” i.e. everybody up to four weeks after the first dose of the Pfizer shot or six weeks after the first dose of the Moderna vax.
This is a meaningless endpoint anyway. The relevant endpoints would be ALL deaths or ALL serious adverse events. I can tell you as a patient it does not matter one whit to me if I die of the covid or I die of something else AT THE EXACT SAME TIME. I don’t care if I have a serious adverse event due to the covid or another equally serious adverse event due to some other cause at the exact same time.
Even if the CDC data showed the shot reduces all-cause mortality or all serious adverse events, that still wouldn’t clinch the matter, because the vaccinated and the unvaccinated are not comparable in every other aspect. There could be any number of potential confounders here, which is precisely why RCT’s were invented – and as we have already noted, the RCT’s were shut down prematurely after Pfizer and Moderna got their (totally meaningless) endpoint of covid infection of any severity – but we’re not going to count infections occurring for or six weeks after the first shot.
Again, as a patient, I can tell you I don’t care if I don’t get covid six weeks from now because I’ve already had it sometime in the previous six weeks.
The mRNA shots are not vaccines as the term has been understood to mean for well over a hundred years. They are therapeutics, which must be administered in advance of infection, with a limited window of efficacy. Is it right to mandate them? Shouldn’t the decision to take them be one that made by the patient and his doctor?
These talks are developing really well.
I like the idea of a Theme, as in Relationship Based Medicine and look forward to the mixed bag of Lawyers and Doctors which might well explain the difficulties that lawyers and doctors have in navigating a Patient Voice, which might be where we come in.
Although touched upon in this talk and GlaxoSmithKline and Study 329, it was interesting that the four questions asked, although good questions, with good answers, were in the main general queries. I can understand that David’s talks contain a lot of information and if you haven’t heard any or many before can stimulate a certain feeling in a certain person.
Let’s hope that some of Relationship Based Medicine hit home with his audience and they become better informed and perhaps a bit more humble when they are out and about working in their field of psychiatry. The data, the ghostwriters, the RCTs, a tangled web which should bring the house down.
Doctors Bearing Gifts, in Calgary; the home of the Calgary Stampede.
I took a flight to Calgary from Vancouver before I lived and worked in the US, just to get a feeling of the place, as it was where my daughter’s father came from and his parents ran a bathroom boutique and the father a mining engineer. He went on to Quebec University as he found Calgary somewhat stifling. In my hire car, I drove through the city centre and out the other side of the skyscrapers and found myself in flat-land for miles and miles and miles. A bit like Las Vegas, as when you come in all you can see is lumps of concrete in a desert.
Doctors Bearing Gifts is a terrific title and it doesn’t matter if you live in a desert, or in a tent in Somalia, I think the over-riding take-out from the talks, is that David knows what he is talking about and I would add that his Subscriber numbers have gone up since posting these talks.
Everyone’s a winner; spread the talks…
So, one ex-FDA senior official describes a Delegated Narcissism effect within the FDA. Of late, the FDA, CDC, WHO and others appear… no thats wrong.. are acting like.. no thats inaccurate too are operating as Medical Dictatorships.
During the Dot Com bubble, I didn’t quite agree with some commentators using psychiatric labels to describe some of these nouveau online webb enterprises, even though I too was at a loss to find suitable descriptive words to describe them (well, not polite ones anyway). However, I’m am weakening when it comes to the whole medical industrial complex and their regulators. I read in the publication Psychology Today an article by Dale Hartley entitled “Are Corporations Inherently Psychopathic?” In it he quotes from Dr. Robert Hare, who being an authority on psychopathy, his thoughts must carry some weight I thought.
Dr. Hare has written this:
“To refer to ‘the’ corporation as psychopathic because of the behaviors of a carefully selected group of companies is like using the traits and behaviors of the most serious high-risk criminals to conclude that ‘the’ criminal (that is, all criminals) is a psychopath. If [common diagnostic criteria] were applied to a random set of corporations, some might apply for the diagnosis of psychopathy, but most would not.”
https://www.psychologytoday.com/us/blog/machiavellians-gulling-the-rubes/201605/are-corporations-inherently-psychopathic#:~:text=The%20fact%20is%2C%20no%20clinical,positive%20or%20false%20negative%20conclusion.
The only thing that appears to influence a psychopath enough to alter his behaviour is when he see that his behaviour goes against his best interests -nothing else. As for collateral damage along the way, well you can’t make an omelet without breaking a few eggs. For the narcissism of psychopathy knows no shame, they’re not wired up to feel such emotions — zero, zilch. And for years, many such healthcare oriented enterprises have promised a few individuals, opportunities to leave by the revolving door and into better paid positions and the inevitable book and TV deals. Any maleficence on the journey will be settled by the organizations at the organizations or taxpayers expense.
I will leave it to the dear reader to contemplate whether we should start calling a spade a spade if and when the hat fits perfectly.
Pfizer Responds to Research Claims
https://www.pfizer.com/news/announcements/pfizer-responds-research-claims
Project Veritas
@Project_Veritas
·
14h
Almost 20,000,000 views
https://twitter.com/Project_Veritas
We might have had Andrew Witty, Patrick Vallance and Paroxetine but what we didn’t have was Jordon Triston Walker, “is this seat taken” …
Relationship Based Medicine, and Pfizer ‘relationship’ based medicine…
‘Never believe anything until it has been officially denied’.
Well done annie. You’ve captured in one post, both halves of what makes that axiom so often true.
Did you watch the clip of James O’Keefe trying to ask the (ex?) director of research and development why he said what he said?
https://www.dailywire.com/news/watch-pfizer-official-physically-assaults-james-okeefe-destroys-ipad-showing-undercover-recordings
P.S.
Certain label’s stick and create life long barriers – as we know– so maybe we ought to use that to our advantage and see if we can get some certain labels to stick to certain organizations which are of a danger to others. What’s sauce for the goose is sauce for the gander.
My guess is by now everybody reading this has seen the Project Veritas videos of that pathetic jackass Jordon Trishton Walker.
“I was lying to impress a date.” That’s the best defense he could think of, when confronted with his own words played back at him.
He claims to be working for a company that is “literally helping people,” so why would he need to lie about what he is doing?
Of course, he wasn’t lying. When he was chortling about the revolving door between the drugmakers and the regulatory agencies that are supposed to protect us, he was telling the truth. And a sordid ugly truth it is.
These are matters literally of life and death, but to him it’s all a big joke. This is a hollow man — one who feels no allegiance to anything greater than himself, who with all his education seems unable to think of any aspirations any higher than the creature comforts afforded by the lavish salary paid by Pfizer (although not for much longer, in all likelihood).
It would be interesting to know his back story. My guess is he is overmortgaged and maxxed out on his credit cards, all in pursuit of new luxuries to titillate his ever-more-jaded palate. Even if he could think of something better to do with his life, I don’t think he could escape the gilded cage he has created for himself.
What will it profit a man if he gains the whole world, yet forfeits his soul?
We’ve peeked behind the curtain and seen the real face of evil. No secret cabals plotting the depopulation of the earth, just a bunch of frightened little men and women with no higher goal in life than meeting their quarterly sales targets.
This guy is the personification of the banality of evil.
Oh, and what do we have here? Looks like those of us on Team Reality were right all along. Wearing a warm moist rag over your mouth doesn’t stop the spread of covid.
Are the people who were spewing out venom at us for the past three years now going to apologize?
Sure, that’ll happen. Right after pigs fly at Mach Three.
https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD006207.pub6/full?utm_source=substack&utm_medium=email&fbclid=IwAR1DuWQPSfDAy71WCUi6i7slIill0QZbgac9RSmqG13-3fEbOjhWne8j_uU#.Y9kxpONYWH0.facebook
Keeping on up…
https://twitter.com/Project_Veritas
“Can you unlock the door” …
Pfizer Director Assaults James O’Keefe & Veritas Staff; Destroys iPad Showing Undercover Recordings
The Sting
https://www.youtube.com/watch?v=u5n7RRKgDog&t=1s
Forbes Hit Piece on Project Veritas is Written By a Scientist Funded by… Guess Who!?
I’ll give you a hint. It rhymes with “riser”…
https://raheemkassam.substack.com/p/forbes-hit-piece-on-project-veritas
I read the Forbes thing a few days ago – what is it with Forbes, and past particles like Matthew Herper…
Martial Arts…
No, Project Veritas Video Doesn’t Prove Pfizer Is Mutating Covid-19, Who Is Jordon Trishton Walker?
Bruce Y. Lee
https://www.forbes.com/sites/brucelee/2023/01/28/no-project-veritas-video-doesnt-prove-pfizer-is-mutating-covid-19-who-is-jordon-trishton-walker/?sh=7d9e41af623d
Miranda Devine Retweeted
Project Veritas
@Project_Veritas
·
22h
Bombshell #DirectedEvolution Story Hits @nypost written by @mirandadevine
“‘There’s a risk,’ he warns. ‘As you could imagine no one wants to be having a pharma company mutating f—cking viruses.’ Understatment of the year.”
https://twitter.com/Project_Veritas/status/1620097900046069761
The Sting, in her tail…
Another Chapter…
Project Veritas
@Project_Veritas
·
11h
BREAKING: @Pfizer Director Concerned Over Women’s Reproductive Heath After COVID-19 Vaccinations “There is something irregular about their menstrual cycles…concerning…The vaccine shouldn’t be interfering with that…It has to be affecting something hormonal…”
#Pfertility
Project Veritas
@Project_Veritas
·
5h
“If something were to happen downstream and it was, like, really bad? I mean, the scale of that scandal would be enormous.”
#Pfertility
https://twitter.com/Project_Veritas/status/1621355064206950406
Third Dr. J. T. Walker/Pfizer-Project Veritas Video
https://rwmalonemd.substack.com/p/third-dr-j-t-walkerpfizer-project?sd=pf
“Could you imagine the scandal?
This was really excellent. I really like the message to the residents about seeing these challengeling cases as research assistants
I’ve written a piece on experiences with SSRIs and their consequences that references your work here and elsewhere. Any constructive feedback and criticism on it from people is welcome.
https://medium.com/@neilbroatch/ssris-marketing-mania-and-suicide-c815011981e1