Following the posting of The Madness of Psychiatry, there has been a flurry of activity in the twittersphere with Louis Appleby, the UK’s suicide czar posting:
What makes adolescents act on suicidal thoughts? New paper shows psychotic symptoms increase risk 20-fold. archpsyc.jamanetwork.com/article.aspx?a…
Others have berated me claiming while I have said we should be aiming at having people on the right treatment for them I am also saying we should have them on treatment rather than not, when in fact what I have advocated for is a recognition of the risks of treatment – be it drugs or ECT or whatever. The right treatment in these circumstances might not involve any physical treatments – a doctor who can’t doctor without drugs is not much of a doctor.
Batman takes on the public health machine
But does treatment come with risks? DSM IV, for all it is castigated, recognizes that antipsychotics cause akathisia and that akathisia can lead to suicide. Is there data rather than just the opinions of a DSM committee? Some of the data has already been posted. The following is from What would Batman do now posted just before James Holmes turned up at the cinema in Aurora (See The Hidden Gorilla).
In the 1950s, the VA hospital system commissioned Norman Farberow to look at rising rates of suicides among veterans. He studied veterans hospitalized for either medical or psychiatric conditions during the periods 1950 through to the mid 1970s. The 3 figures below bring out the findings.
Figure 1 shows a set of fluctuating suicide rates year on year for veterans admitted to medical beds. The rates are higher than national suicide rates but these rates and their fluctuations are in keeping with what might have been expected in a set of younger men. The increases in the late 1950s and early 1970s may mirror the effects of the Korean and Vietnam wars, or perhaps other social factors or they may be entirely random.
Figure 1
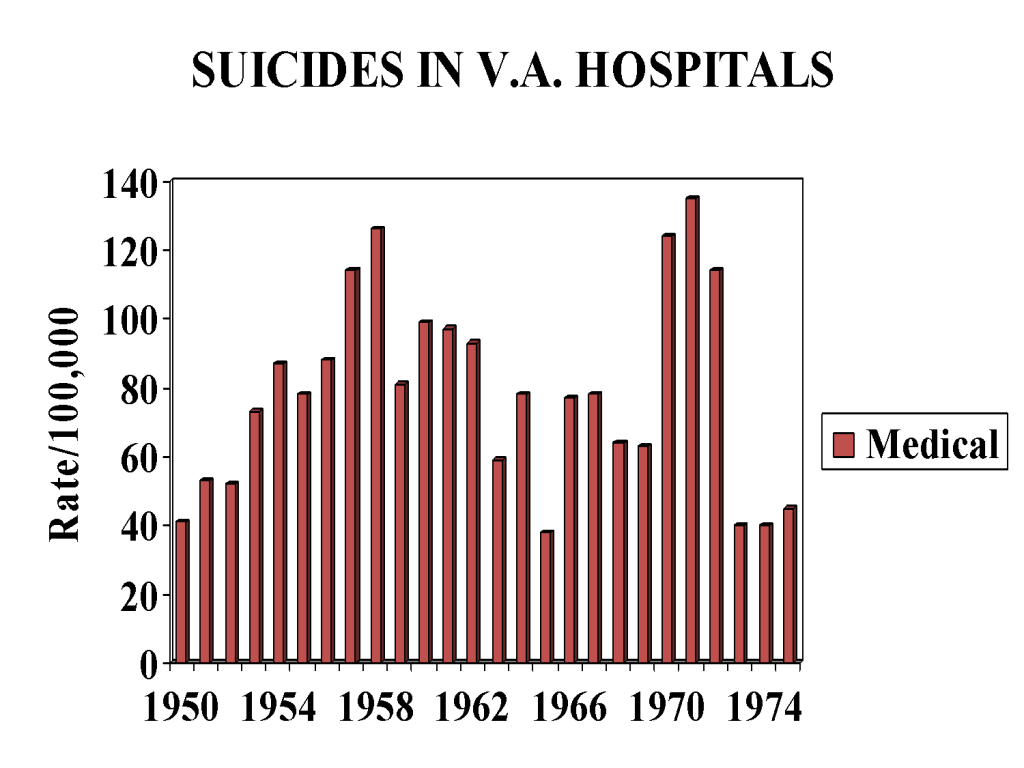
Figures 2 and 3 are strikingly different to Figure 1. Figure 2 does not show the expected fluctuations linked to social factors or any randomness. It shows a steady rise in suicide rates in those who have been hospitalized for a mental condition. Until 1955 the rates are identical to the rates found in those hospitalized for a general medical condition.
But as of 1955, they start climbing in an uninterrupted fashion. The rises and falls we see in Figure 1 that might or might not be linked to social factors such as the Korean war are not there. This can be seen clearly in Figure 2 when the two sets of figures are superimposed and again in Figure 3 which show admissions to psychiatric beds on their own.
Figure 2
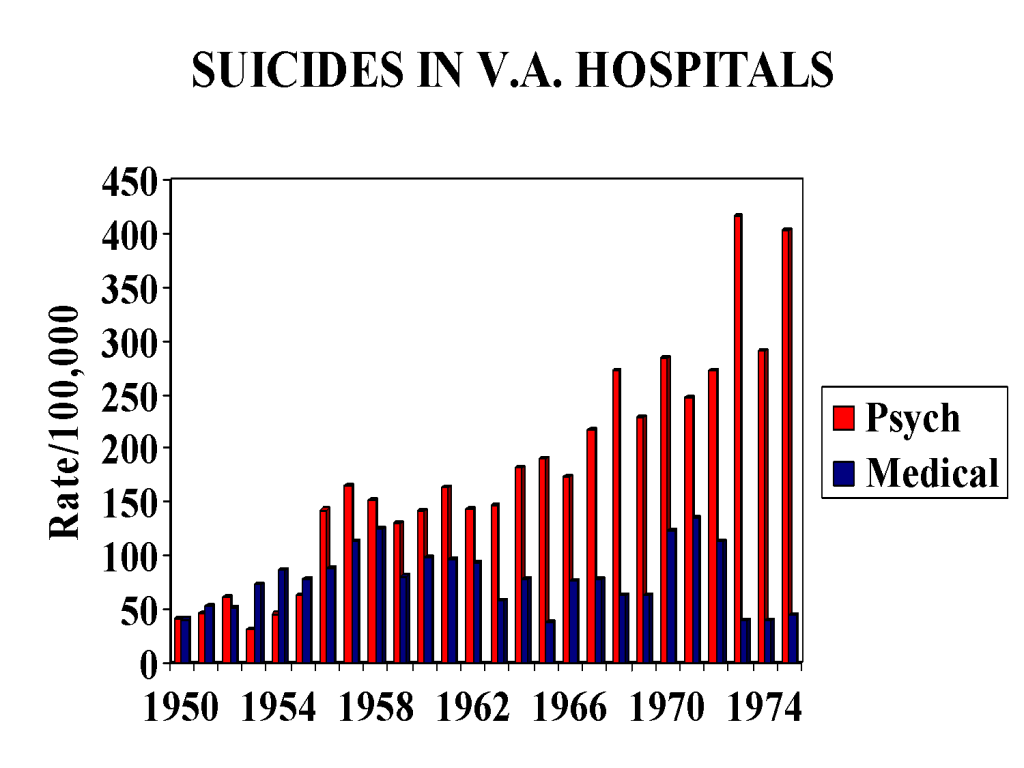
Figure 3
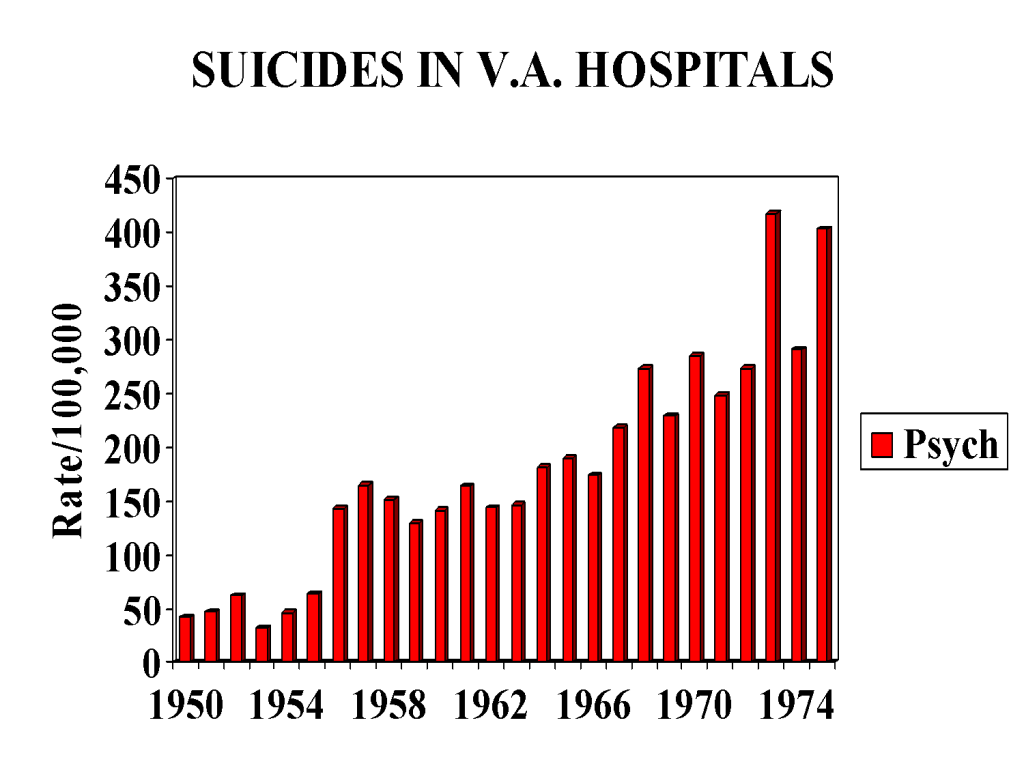
Why the bifurcation in 1955? This was the year of the introduction of chlorpromazine. Year on year after 1955 a greater number of tranquilizers (antipsychotics / neuroleptics) like chlorpromazine were consumed by veterans with mental health problems as an ever greater number of these drugs were marketed. These drugs were given to veterans who were depressed, anxious or psychotic – they were not as might be thought now restricted to veterans who were schizophrenic.
The question for Batman in the face of rising suicides in the US military, what would he do now.
What’s with the question marks?
In 2001, Khan et al had an article in the American Journal of Psychiatry which showed the figures for Risperdal, Zyprexa and Seroquel below. Intrigued by the high rates of suicide on Zyprexa, but even more so by the question marks where the missing data should be, I checked the FDA reviews for this drug and found unlike other reviews for other drugs the data was indeed missing. I have added extra data for Sertindole, and Geodon.
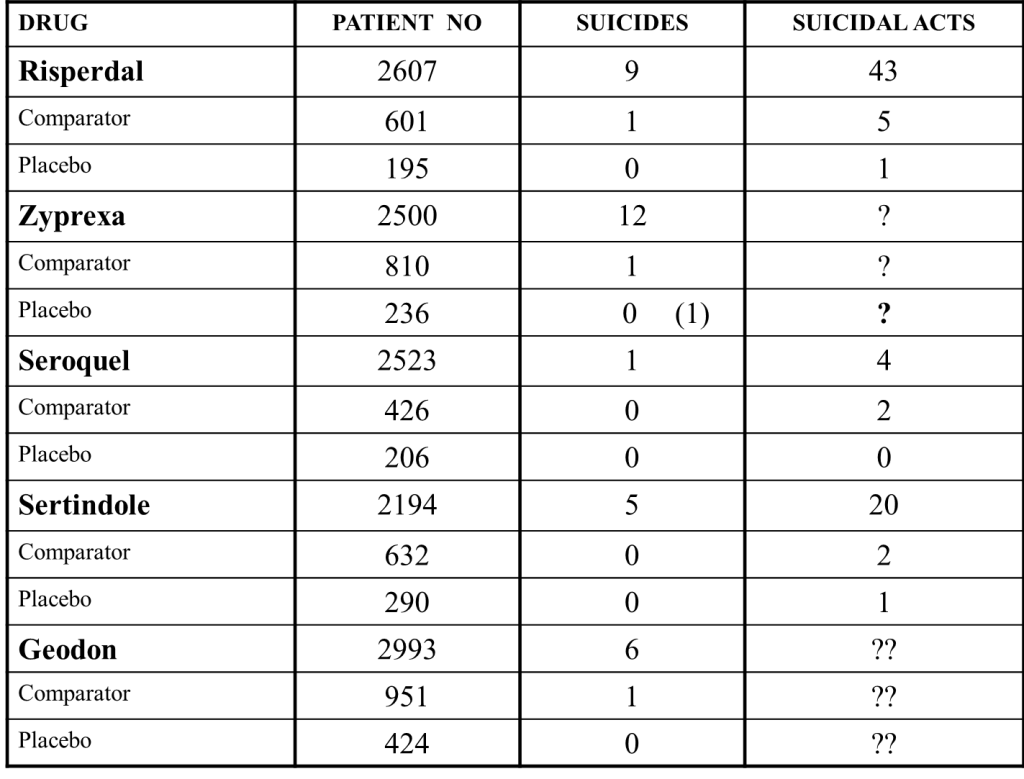
What if we tell him we didn’t collect the data on suicides?
I wrote to Lilly asking for the data and had the following response:
Dear Dr Healy
Thank-you for your letter concerning suicide attempts in clinical trials with olanzapine, which was forwarded to us by Harry Owens. I am sorry you did not find our previous letter on this subject helpful.
Your question was referred to our parent company in the USA, but unfortunately the specific data you requested are not available.
Yours sincerely – Dr A Simpson, Medical Director
The previous letter had intimated the data didn’t exist, which was scarcely credible. This was a time when the company according to documents later obtained under FOI were doing everything they could to get me or my junior doctor to prescribe Zyprexa, not showing me the data made it impossible to prescribe their drug.
Informed consent and Zyprexa
I wrote to the then Minister for Health Alan Milburn to see if he could help as he had responsibility for clinical trials in the UK, which could not both involve Zyprexa and informed consent if these data were not available. I never got a reply. Not even an acknowledgement. This was a time when Lilly it is rumored were threatening to pull out of the UK if Zyprexa were not written prominently into the Guideline for Schizophrenia, which was the very first NICE Guideline.
I later got to see the data. The number of suicidal acts on Zyprexa was higher than the number on Risperdal.
These data do not include suicides and suicidal acts that might have been triggered by withdrawal. In a 1993 FDA approval review of Risperdal, Andrew Mosholder, one of the agencies reviewers, notes that:
“The sponsor reports no instance of risperidone abuse or dependence. Withdrawal phenomena were not formally assessed after patients discontinued risperidone. Several patients committed suicide within one month of discontinuing risperidone, however, it does not seem reasonable to attribute this to withdrawal, given the absence of other indications of a risperidone withdrawal syndrome and the fact that schizophrenia is known to be a risk factor for suicide”.
The Madness of Psychiatry shows this is a totally unwarranted assumption. There is a compelling case antipsychotics cause dependence and withdrawal (See Tranter and Healy 1998).
The data
These clinical trial data are ambiguous. They are not good quality data. There is no adjustment for patient exposure, and in this case some adjustment is called for but there is no way to undertake it. There should also be data for suicidal acts during the withdrawal period but these data are not included.
As they stand the data show a statistically significant increase in risk. This doesn’t mean antipsychotics cause suicide, it means that in these trials they caused a significantly greater number of suicides and suicidal acts than happened on placebo.
Although the data are poor, you might have thought journals would be interested. Far from it. They are not prepared to publish, even though this is the best we have and there is not a journal editor who does not trumpet clinical trial data as the gold standard.
In fact clinical trials are close to useless when it comes to suicide. It is easy to design a study of a drug known to cause suicide that would show a reduced rate of suicide compared to placebo (See Healy 2012). Clinical trials function instead for public health officials and journal editors as a bureaucrat’s tool to avoid exercising judgement. When they pose problems like the data here do – better they remain unpublished.
The bureaucrat will see you now
Louis Appleby and other officials take a public health or vaccination perspective. This is essentially anti-medical when it comes to drugs. It accepts casualties without attempting to mitigate the risks. There is not supposed to be any questioning of drugs any more than there might be of vaccines. When officials like this talk about benefit risk ratios, they mean benefit on a public scale that includes job creation or the relocation of a company like Lilly out of the UK rather than the good of patients. The benefit is moreover ambigous – Zyprexa would in fact have an effect for GPI – dementia paralytica – tertiary syphilis, but using it might get in the way of us discovering that in fact penicillin would be a real benefit. The language has been perverted – an effect that suits some interest groups has been termed a benefit when it may well not be for many.
If bureaucrats of this type had run the automobile industry from the start they would never have allowed brakes in cars – and it would be close to impossible to persuade them that good drivers can in fact drive quicker if they have brakes than if they don’t. If you want a good driver or a good doctor these days, well now that’s getting harder and harder to find here in North Korea.
There is no data on clozapine?
La Reine Margot post, coming soon, will have some
Hi David. Thank you so much for this article and data you have supplied. I am on Relprevv and your data supports the way that the drugs are making me feel. When I try to explain to the Doctors that I feel suicidal on the anti psychotics and anti depressants it feels like they don’t believe me and tell me that I am feeling suicidal for other reasons. But I have had 1 trial period were I was off the drugs and I notice a considerable change in mood. It was like going from sad to happy, and when they put me back on the drugs from happy to sad.
I just have a question regarding the data provided under the “What’s with the Question Marks?”. Can you please give the numbers of how many patients were in the trial for each drug?
I’m not a doctor, but I have been studying the effects these hard core drugs have on people and the correlation between them and suicide and suicidal thoughts and behavior. It’s hard to get the numbers of people that have been harmed while taking these drugs since there is a huge effort to hide this information from the public. The pharmaceutical industry is the most powerful and lucrative in the world.
That being said, please research a safe and natural way to maybe ween off anti depressants with advice and help from natural heeling doctors, not ones that receive incentives and kick backs from the dangerous drug companies making the very drugs that they are pushing.
If the BMJ and other psychiatric and medical journals are unwilling to publish because ‘it knocks medicine on it’s head’, because they haven’t yet arrived, or even set off on the journey, then might I suggest a New World Medical Journal appears on the market.
You could fill a magazine with explosive research and data and stories, from Sweden, from New Zealand, from Canada, from the Indians who are being manipulated into clinical trials organised by GSK and dying needlessly, as exposed in Newsnight this week.
Your advertising revenue could come from thousands of areas based on the premise:
Don’t take a drug unless you realise the Rxisk
Holidays, Travel Agencies, Property, Tourism, Natural Health, etc.etc.
Every time I read some spurious article in the newspapers about drugs and how they can help, ie ssris I am amazed that we cannot retaliate at all. In fact, I think it is pretty irresponsible of newspapers to publish articles like this at all.
All articles should contain facts and figures and risks, not all the benefits, as they do.
I always think that every drug article I read should have a response article from an eminent professional. It is all so damningly misleading and dangerous.
An example, today, front page, Daily Express, Statins in new health alert!
This is not new, news. This statin saga, is like the ssri saga, just as dangerous, just as misleading.
You cannot have half the world saying ‘take it’ and the other half saying ‘don’t take it.’
It is medically insane. Everybody wants evidence based medicine, the majority think that is what they are getting.
“A doctor who can’t doctor without drugs is not much of a doctor.” Now, that’s a breath of fresh air! A lot of docs have lost any sense of that — and I’ve met a few who are more like Pez dispensers, sad to say.
If these trials were run mainly on people diagnosed with schizophrenia – and if those people don’t innately have a high risk of suicide – then we are in real trouble. Because these drugs are being doled out more and more to people who aren’t schizophrenic, but whose other problems do put them at high risk of suicide. They could be triggering even worse suicide rates than shown in these tables, and the problem could hide out in plain sight.
Take veterans. The number of vets (and active-duty soldiers!) being prescribed Seroquel in particular has risen high enough to grab the attention of the Joint Chiefs of Staff. Most of them are getting it for post-traumatic stress disorder, and in high doses. Others are getting it as an “add-on” for depression or as a sleeping pill of last resort for folks in chronic pain from physical injuries. The suicide rate among vets is horrendous as it is. If 10% of that, say, were a consequence of the drugs they were on, would anyone catch it?
I also see a lot of civilians in chronic pain getting Seroquel or Risperdal as a sleeping pill – including patients with serious back injuries, who end up adding obesity and diabetes to all the other stresses on their bodies. Any suicides there will be chalked up to the physical pain, loss of a job, money woes, etc. I’m sure.
Among those getting antipsychotics as mood-stabilizers or antidepressants are many people diagnosed with “borderline personality disorder” (BPD) and similar problems. I spoke with a social worker recently who runs groups for young adults with BPD. By definition, they have turbulent emotional lives and respond to distress with impulsive actions, including suicide attempts, cutting, drinking & drugs, etc. She said a lot of her clients take Abilify, but they don’t complain about akathisia so much as weight gain. Patients with a lot of “drama” in their lives to begin with may not be able to pinpoint the cause if they begin feeling worse – and their treaters still less. Abilify could turn out to be the riskiest drug of all.
A gorgeous lady, from an affluent background, this week. killed her two babies, because she stopped her an anti-depressant.
It is not now rocket science to see why this happened. Luckily for her, the judge concluded that she had ‘forces’ beyond her control. How will this woman ever get over this. She will receive further medication, without a doubt, and if it is ever recognised that her drugs were to blame, how will she ever come to terms with it.
Well, she won’t. Her life is now so severely damaged, from an ssri, that all the evidence in the world won’t bring her babies back.
We know, what it is like to have ‘an episode’ from these disgusting drugs and I can only feel overwhelming sorrow for the woman who ‘lost it’ after stopping her drug.
I honestly don’t know how these pharma executives sleep at night, crowing away at their awesome and spectacular ‘deviations’ to the mind of individuals like this poor lady.
It is difficult for some people to understand how “crazy” psychotic drugs are – some weeks ago a doctor I spoke to re feeling down “and stuck in the past” and wishing to “go back and change things” told me I was just depressed – he prescribed sleeping tablets and Lexapro – I did not know at that time that Lexapro was a psychotic drug nor was it explained to me – Wishing, wanting and praying to feel better and be an obedient patient I took Lexapro for five days – (I stopped because of the feelings of feeling too high – to feelings of exhaustion. Within a short time after stopping them I knew I was reacting more angrily to situations that arose especially in disagreements/arguments with a close friend – the anger was frightening – whilst I would be upset later for reacting so angrily there is something I find difficult to work out – it is a profound feeling of detachment from the “episodes” and an overwhelming feeling of “some part of me lost”. It is a frightening lonely road.
In 1970, my brother was killed in a tragic road accident in Saudia Arabia. He worked for Marconi. He was twenty five years old.
My parents and I were stricken with grief, My lovely dad was put on valium, which he remained on for twenty years. As his child, I was constantly aware of my dad, taking a pill out of a wee box, but I didn’t know anything about pills.
My mum was put on Ativan for her grief. She, also, was on this for years. When she stopped taking Ativan, she had a complete nervous breakdown. It was appalling, but, despite that, she got over it.
Dad stopped taking Valium, when he retired, and never looked back. He was a better dad, after valium, and, mum, off her Ativan, was a fabulous mum.
I went to see a psychiatrist in 1988 and he gave me Lofepramine. Within a short space of time, I had suicidal thoughts. To his credit his decided not to continue the pharmapyschology approach and let me get on with my life.
Another crisis, airline pilot boyfriend drunk, and Sunday Mail arrive on my doorstep, and it is all over the newspapers.
What to do, give me Seroxat to cure my ‘problem.’
Some absurd dimwit, deep in my medical records, said I had ‘genetic loading’.
This man, said I had genetic loading, I ask the question, why was my dad put on valium and my mum put on ativan, for years and years, and we came through it but then, astonishingly, I get Lofepramine and Seroxat from which Lofrepramine caused ‘suicidal thoughts’ and Seroxat caused ‘suicide’.
I also had a distant cousin, who commited suicide, because his wife left him with the babes. This was also given cause of concern of ‘genetic loading,’ He was a police sergeant, he was stricken. It was nothing to do with me.
What is it about psychiatrists, wanting to medicate, and not listening, or even attempting to realise, that Valium, Ativan, Lofepramine or Seroxat and, of course, all the other humungous drugs, do more harm, in disablement of the’ human right to a normal life.’
My dad died of an aneuryism, but happy and fulfilled, off valium, my mum is now 92, bright, alert and fabulous.
The damage these drugs cause is beyond redemption.
Thank the lord, drugs are no part of our lives anymore and we can get on with living, not medicated.
‘Genetic loading’. I can’t quite grasp what his point was.
I wish to know why when I withdraw and stopped too suddenly risperadon I suffered teeth shattering AND NOT BEING ABLE TO TALK FOR 2 HOURS. I would like a copy of evidence of rebound effects for risperadon and to know if the symtoms are actually a cause of the tablets and not my illness ( psychosis)
The withdrawl from rispiradon was too sudden and I had “weird sensations in my head ” (hARD TO DESCRIBE). David kealy can you supply me a copy of rebound effects of rispirodon please so I can present evidence to the prescriber the psychartrist. who just passed me on to his senior doctor. I have been passed around three doctors now with no answers. They are just saying all my symtoms are psychological. I actually am more suicidal in that I have physical symptoms and my fear of not being understood is ever more pressing.Can you please help.
Yes indeed a brave and good doctor.
Ten years ago, in an Irish mental hospital, I was put on respiritdol immediately after telling the psychiartrist that I had been smoking grass for a few years to relax, but now wanted to stop.
Trusting in her judgement I took the prescribed dose everyday for two months, by the end of which I was just dreaming of ways to kill myself.
When I used to see that someone on TV had died, I would say, ” You lucky B*stard”. The compunction to kill myself was far greater than any desire I felt for grass.
Eventually I tried to OD myself with a month’s supply of pills, luckily my girlfriend came home and got me to the hospital on time.
I was never suicidal before taking those pills and have not been since.
What makes me really angry is that, the seniors in our society, never stand up for the truth in relation to this problem, they just are too indifferent and lazy to look up facts, make enquiries and demand some answers from politicians.
but sure what can you do?
big pharma pays the pols to do their bidding and sure who can blame them?
souls don’t come free ya know!!
Well id say its pretty obvious that antipsychotics would cause a lot of suicides. They are semi-permanent dysphoriants after all…
Ronnie, I know what you mean. Ive been on and off these drugs for many years and I envy those that are dead.
I have become increasingly ill after I have been given antipsychotics for PTSD. After withdrawing antipsychotics I got my first psychosis. At the moment I am severely suicidal because I am stuck on antipsychotics. I can’t get off anymore, because they made me chronically psychotic. But life on antipsychotics is not worth living – they ruined my life. I can’t feel love, joy, happiness, faith, anything that makes life worthwhile. Thank you for bringing a subject like this to our attention.
I agree Seroquel can cause suicidal behavior, even in older people, and wonder how to do something about it to warn people. Your writing about veterans and suicide while taking Seroquel is very revealing.
I was placed on Xeplion/Invenga sustenna for 6 months and I got a lot of adverse effects. I am definitely suicidal because they took away all my sleep which will kill me in a horrible way. I don’t know where to ask for help