Editorial: We interrupt the Go Figure series of posts to return for two posts to Study 329. We will then return to Go Figure.
All the fuss about Study 329 centers on its 8 week acute phase. But this study had a 24 week Continuation Phase that has never been published. Until Now.
We might have Marty Keller to thank for this Continuation Phase. His big deal was the long term treatment of depression. He made his name on the back of claims that depression was more chronic than people thought, and that treatment might have to carry on much longer than had been thought. Or perhaps industry made his name for him as this was such an appealing idea for them. He rapidly became one of the go-to key opinion leaders for antidepressants, along with Charlie Nemeroff in the US and Stuart Montgomery in Europe.
Rhode Island, and Brown University, must have looked Providential to Pharma in the early 1990s. Peter Kramer of Listening to Prozac fame was there too, also advocating pretty well permanent treatment for people who might be somewhat less than entirely chipper 100% of the time. Well would you take off a pair of spectacles if your sight was crisper wearing them? Why treat a drug any differently? We need to get over these Calvinist hang-ups about feeling better on medication, maybe even feeling better than well.
The continuation phase
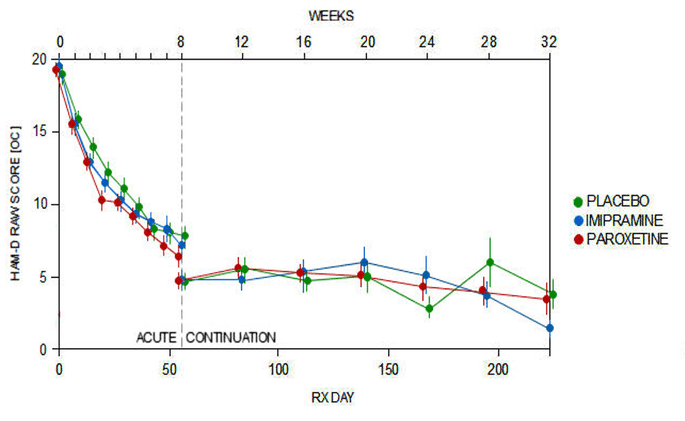
The Acute Phase of Study 329 ran for 8 weeks – slightly longer than usual because the worry even before it began was that it was going to be difficult to show that treatment worked. But then after that the children could enter a further 24 week continuation phase. The idea was to see how ongoing treatment shaped up. The continuation phase was never published. Never even talked about.
To coincide with the Autumn Equinox, the International Journal of Risk and Safety in Medicine has published Study 329: The Continuation Phase.
The full text is available on Study 329.org, along with the reviews from the Journal of the American Association for Child and Adolescent Psychiatry (JAACAP). Click HERE.
JAACAP is not IJRSM – what’s up?
The original Keller paper was published in JAACAP. The former editor Mina Dulcan and the current editor Andres Martin and the American Association for Child and Adolescent Psychiatry were lobbied heavily over the years by Leemon McHenry and Jon Jureidini, trying to get them to do the decent thing and retract the paper. In vain.
So it seemed like a good idea to send Study 329 The Continuation Phase to JAACAP first. We were pretty certain they would refuse it. But they needed to be given the opportunity to dig a deeper hole. And they did. The reviews are published on Study 329.org.
JAACAP
The primary theme of the reviews is there is too much data here, tell us what it means. Interpret it for us, and then we can tell you whether we agree with you or not. The paper may in fact have too many tables and figures but the key message behind Study 329 is that there is no authoritative interpretation. A lot hinges on almost arbitrary categorizations of the data. You the reader need to be able to play around with the data; you may well spot things others have missed. When it comes to adverse event data in particular there is no expertise. Things need to be picked over.
Off a cliff
There are some things about the Continuation Phase that are almost too shady to pick over too much, and we did not spend time on them. At the end of the Acute phase, for instance, there was an option for patients to continue into the next phase. It’s easy to understand why people doing poorly in the trial might have opted to drop out at that point. Some people doing well can also have been expected to drop out. What is harder to explain is why most of those doing well who dropped out were on placebo. A mixture of reasons were offered such as non-availability of trial medication but two thirds of those dropping out while doing well were taking placebo. This does not seem random. From GSK’s point of view having a large number of people doing well on placebo enter the continuation phase would not have been ideal.
The findings
Despite the placebo dropouts, there are more data to pick over in this study that for any other study. The data are not flattering for paroxetine – Paxil / Seroxat.
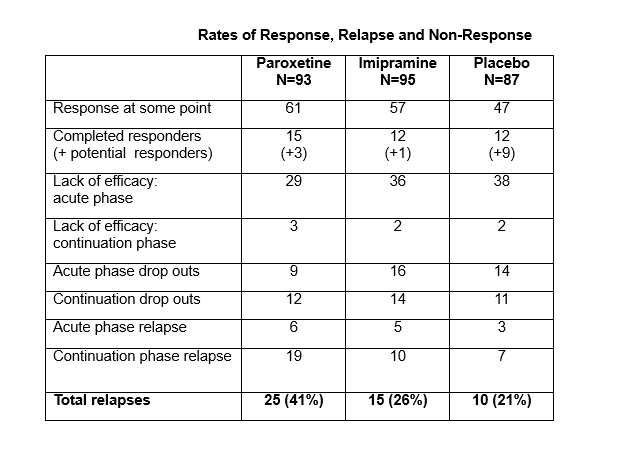
The relapse rate on paroxetine was higher than for imipramine or placebo. This relapse rate makes paroxetine a Gateway drug. People taking it end up worse off and their difficulties are interpreted as further evidence of illness, and in the real world this is likely to lead to additional diagnoses and additional treatments.
As the Efficacy data above shows, there is no evidence that patients given paroxetine show any benefits compared to placebo over the longer run – even when the deck is stacked against placebo by eliminating many of those who are taking it and doing well.
The suicide data also continued to be a problem into the Continuation phase as the graph below shows.
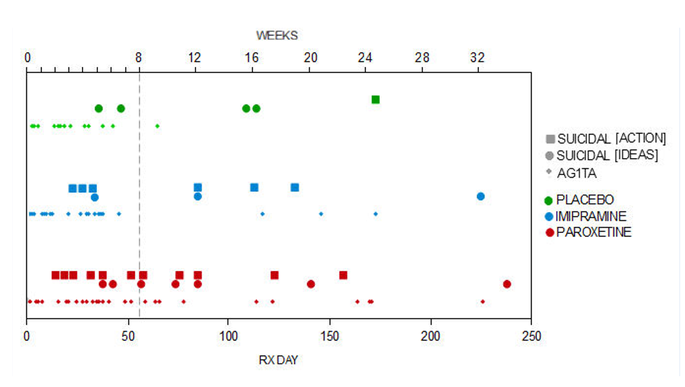
The word AG1TA here stands for agitation.
But there was another even more interesting finding in this paper which will feature in next weeks post. If you download the paper you will likely spot it. Surprisingly in the light of the finding, or unsurprisingly given the finding, the reviewers paid no heed it.
To be continued…
At the Aspen Institute, the discourse suggested PC (pharmaceutical companies) might like to pre-empt dis-ease by R & D; by having the right drug available at the right time in case of a mass epidemic.
Designing Depression as a Disease and by developing drugs to combat disease – this extended trial of 24 weeks threw up the ‘Sans Andreas Fault’
#6
A refined and articulate and crucial study by RIAT Authors comparing Paroxetine to Placebo.
The designation of which was an add-on to the infamous Study329, as published by Keller et al and aided by Nemeroff/Paxil workshops/fees/disgraced
The Authors have shown through careful scrutiny and challenging research, how important is this re visitation; the epidemic of world-wide deaths from Paroxetine.
It is our job and our conscience that all teen deaths and adult deaths by Paroxetine are queried by all means possible.
With this in mind, the RIAT Continuation Study will not be published in JAACAP as JAACAP see no reason to halt future deaths from Paroxetine.
Deaths and ADRs from Paroxetine are streamed regularly and consistently in the UK…Canada/USA/Switzerland/Eire..
To be ignored…
We have also looked at all behavioral adverse events in each phase (Table 11 and Fig. 4).
These events include: agitation, aggravated depression, akathisia, abnormal dreams, depersonalisation,
disinhibition, feelings of despair, hallucinations, impulsive behavior, negative thoughts, neurosis,
paranoia, psychosis, suicide attempt, suicidal ideation, suicidal gesture, self-harm, and self-injury.
“Classifying “akathisia”, “aggravated depression”, “Abnormal dreams”, and “depersonalization”, as a
potential suicidal event (per Appendix 3) is highly questionable”
http://study329.org/the-data/
In 2004 as part of the resolution of a fraud action taken by New York State, GlaxoSmithKline agreed to post the data from all their studies on the Company Website. They posted lengthy Clinical Study Reports (CSRs) for all the pediatric antidepressant studies (Paxil).
http://study329.org/wp-content/uploads/2015/09/Full-study-report-continuation.pdf
How appropriate that Professor David Healy has a prestigious talk today at QU on the Law and Medicine
http://psychiatry.queensu.ca/assets/grand-rds/2016/Speaker_Healy_20160928.pdf
Here’s hoping for the video link..
‘GlaxoSmithKline now faces Trial for Fraud’
Reframing the essential Video: Jeremy Vine/Seroxat/Shelley Jofre/BH Lawyer/1BOM/consolidating the Data..
Panorama/Confidental archives..
https://www.youtube.com/watch?v=_0ffzsrDkSQ
https://truthman30.wordpress.com/2016/09/28/hey-emma-walmsley-as-a-mother-of-4-what-do-you-think-of-seroxat-study-329-and-the-paroxetine-child-suicides-does-this-make-you-proud-to-be-part-of-gsk/
Mickey
September 29, 2016 | 12:59 AM
1bom
I think you’re referring to Table 13. The graphic posted at the end here clarifies that table. The events you mention are what I called “AGITA” or “Agitation” in Healy’s post. The concrete SUICIDAL ACTS and SUICIDAL IDEATION are clearly separated out. The explanation of the “why” of Table 13 is in the second paragraph under 2.7 Analysis of safety data. Obviously, the hypothesis here is that an internal state of agitation lies on a continuum with the more ominous suicidal ideation and behavior, and we were casting a broad net to collect that information. Looking at the graphic, the three things do appear to “run together.” We weren’t trying to pad the numbers. We were intending to explore that association…
http://1boringoldman.com/index.php/2016/09/28/study-329-something-new/
http://www.madinamerica.com/2016/09/study-329-continuation-phase/
Memory jog..
Are we expecting another re re response
*This is widely known and clear warnings have been in place on the product label for more than a decade. As such we don’t believe this reanalysis affects patient safety*
http://www.gsk.com/en-gb/media/resource-centre/paroxetine/gsk-response-to-paroxetine-study-329-reanalysis/
GSK responds to research group’s paroxetine study 329 reanalysis published in the BMJ
We were able to help this team to carry out their reanalysis by providing access to the detailed data from the original trial. This reflects our commitment to data transparency – we publish the results of all our studies regardless of whether they are positive or negative and we are the only pharmaceutical company to be part of the AllTrials campaign. We have also led the way in giving external researchers access to the very detailed patient-level data behind our studies, granting access to more than 50 research teams around the world so they can independently use our data in their research.
Importantly, the findings from this team’s analysis appear to be in line with the longstanding view that there is an increased risk of suicidality in paediatric and adolescent patients given antidepressants like paroxetine. This is widely known and clear warnings have been in place on the product label for more than a decade. As such we don’t believe this reanalysis affects patient safety.
This particular trial, which was conducted in the 1990s, was included in detailed reviews carried out many years ago by regulators and by GSK which identified the increased risk. It’s something the medical community and regulators are aware of.
Learn more about our commitment to data transparency.
Signed up..
http://1boringoldman.com/index.php/2016/09/29/clinical-trials-an-act-of-congress/
Graph 1: My doctor told me, in about 1999, that I’d have to wait 4-6 weeks for paroxetine to have its full effect. I also know other SSRI’s are said to have a delayed onset of effect. And the curves are similar to placebo, I interpret the inital phase as placebo. Same goes for continuation phase, no distinct improvement can be found compared to placebo.
Graph 2: Indeed not flattering at all. And if this is to be considered a longer (than normal, but still short in my mind) trial, then it is amazing how anyone can bypass the obvious worsening for a greater portion of the paroxetine group than both the comparison groups. If anything, this should have been interpreted as atleast reason for further investigation. Why do paroxetine trigger almost double the rate of relapse?
Graph 3: I can only shake my head. Remember that any green marking is a human response to its state and health. Both blue and red are after intervention. It speaks for itself.
We cannot accept that a graph shows, without a doubt, that pharmaceuticals lead to suicide, thoughts of suicide, and agitation. Agitation means that sooner or later, this person has an increased risk of becoming violent. Even if this behaviour and thoughts can be found in a patient without drugs, we cannot risk that the human intervention is the cause. Suicides has always plagued humans. This is not a way to reduce such.
Disclaimer: I have only read this post and the graphs here on this page!
Gut-wrenching and true……with thanks to Katinka, amazing children and all the others speaking out
My new 10 min campaign film re dangers #antidepressants #akathisia
Katinka B Newman @pillthatsteals 13h13 hours ago
The Pill That Steals Lives
https://www.youtube.com/watch?v=eEeJtRB5hqg&feature=youtu.be
Thanks Annie.
Katinka – this is absolutely brilliant! Many of us have wondered what the best way is for moving things forward and grasping the public’s attention – you have shown us the way. There cannot be many better ways than to show the reality of what can happen by means of film. That you actually had a snapshot of ‘the drugged you’ as well as the ‘real’ you and the effects on your children is quite amazing. Their memories of that time and their comments will awaken the reality of what can happen in a new, younger generation which, to my mind, is so important. Thank you so much.
Martin Keller, Principal Investigator of Controversial Paxil Study, Leaves Brown University
By Alison Bass
Featured Blogs September 5, 2012
http://www.madinamerica.com/2012/09/martin-keller-principal-investigator-of-controversial-paxil-study-is-retired-from-brown-university
September, 18, 2015 | 23 Comments
Most of us were here..
https://davidhealy.org/study-329-mk-hk-sk-and-gsk/
I would like to understand why suddenly at age 18 it is safe to take Seroxat, whereas up to that age, classed as ‘child’ or ‘teen’ it isn’t. Does the brain suddenly change on one’s 18 the birthday? We always understood that young brain is not fully formed till age 24, is it something to do with parental responsibility changing at 18 when you become an adult.
Our very slim 21 year old took it for first time then and was never the same again. He stopped it after a few months and had such terrible panic attacks he had to leave Uni in his second year. We and he never guessed it was the drug, His girlfriend at Uni was Portugese. Her mother was a pathologist. In 2002/3 she rang me to warn me about problems with Seroxat, so she must have had good medical info over in Portugal. We tried to talk this through with his Hereford psychiatrist and he became angry and stopped us visiting our son in hospital, who was by then suicidal and saying he had to go ‘cos he was needed in heaven, as there was special work there for him to do’. Even MIND would not help us raise the subject with the arrogant psychiatrist. Meantime the pathologist in Portugal was desperate for him. He had been all set for a first class degree. He had worried about his acne, sure, but was never ever suicidal like he became on both RoAccutane/ isotretinoin and Seroxat.
Time and again, a young person becomes uncharacteristically ill in their thoughts on this drug but the parents are not listened to. Our son kept blaming himself for his suicidal thoughts and the medics accepted his own verdict, but it was obvious to us that he was out of his mind so that’s why he wasn’t making any sense. I just don’t understand why the Portugese medical people were ringing alarm bells in 2002/3 and in UK, we were not.
H
The effects of the drug are the same in all age brackets. There is nothing in particular about youth.
DH
It is ‘patently’ absurd..
http://www.telegraph.co.uk/news/uknews/1432640/Seroxat-can-be-a-danger-to-under-18s-doctors-told.html
http://news.bbc.co.uk/1/hi/health/2975844.stm
http://www.scotsman.com/business/companies/tech/gsk-faces-fresh-seroxat-woe-1-651442
Telegraph:
However, there is no evidence that it is harmful for adults, the medicines and health care products regulatory agency (MHRA) said. The agency warned people not to stop taking Seroxat, also known as paroxetine, without consulting their doctor. Coming off the drug quickly without medical supervision can trigger unpleasant and potentially dangerous side effects.
BBC:
The sentence “Remember that you cannot become addicted to Seroxat” has been removed.
Instead, patients are warned not to stop taking Seroxat unless they are told to do so by their doctor and told if they do stop taking it suddenly, they are likely to suffer side effects such as dizziness, agitation or restlessness and nausea.
Scotsman:
A GOVERNMENT agency is today expected to deliver a damning verdict on GlaxoSmithKline’s widely-criticised anti-depressant Seroxat.
Two years ago, a jury in the US awarded US$6.4m (3.9m) to the family of a man who murdered his wife, daughter and grand-daughter and then committed suicide in what the family claimed was a reaction to the drug. GSK denies the connection.
Thanks DH for clarification re age limit for young Seroxat-takers. Obviously my question was put ‘tongue in cheek’, and as Annie states in her follow up comment, the age stuff argued by GSK is patently absurd. Our son’s body weight age 21, being of slim build, would have been no more than that of the average 17 year old. So physically he could have been classed as a teenager.
Thanks Annie too for all those interesting references.
The more I read about Seroxat, the more frustrated I feel. I can begin to sense physical almost panic-like symptoms myself now, which I guess come from the powerlessness I remember when trying to save our son and be listened to; and it’s maybe good for me to experience this kind of anxiety/panic now because it makes me realise a little of how anxiety sufferers feel, and how EASY it would be, and how understandable, to go to the GP for a comforting pill to ease the pain.
The only thing that helps is to talk it out, or to write it down. Knowing what I do now about medication, I would be very wary of going to the GP before exploring other options to help me. But one can see how natural it would be for most people who haven’t read RxISK reports, to ask for medication to help, which could well leave them in a much worse situation.
Interesting comment re: your son’s build and bodyweight etc. Our son, when put on Seroxat and had the devastating reactions to it, was also around the same age and build as you describe. I suppose it would follow that the dose prescribed should reflect the person’s build etc. – but in our son’s case, that was certainly not taken into account as he was put on 30 mg. daily from day one!
I also note your comment about your own feelings of anxiety etc. I share those feelings – have done for the past fourteen years and more! W e are the lucky ones, I feel, we can put our thoughts and worries into words and release that pressure in ourselves by doing so. On the whole, those who suffer severe anxiety find it hard to talk about their feelings, which, as you say sends them on their way to their GP to be presented with a prescription. Why, as a nation, have we become so scared of our natural emotions? Show that you are happy, sociable and contented and all is well – dare to be unhappy, miserable or lonely and you are seen as being in need of ‘a happy pill’. Why can’t we accept that to experience the whole spectrum of emotions is the only way to become fully-rounded human beings? That we cope through ‘knowing’ and not through ‘masking’ the depths of our emotions.
Dear Mary, I’m so sorry to think of you suffering this way for so many years and still doing so. You are bang on right when you say it’s easier to deal with by ‘knowing’ not through ‘masking’ the depths of our emotions.
I think a lot of anxiety gets triggered by fear of the uncertainties in our lives, worries about how long our job will last, then how will we pay the mortgage, or our health, are our symptoms indicating some hidden major serious illness, etc. When these worries go round and round in the mind, especially at bedtime, one feels worse and worse and it’s difficult to sleep. Insomnia then spirals us into more anxiety next day.
I know it’s not fashionable to say this, and lots of people will no doubt groan to read it, but back say 100 years ago, a high percentage of the population believed in some enveloping loving power which watched over and cared for each of us. It could be God, angels, nature, whatever, but they felt they were all part of some grand design, and that somehow we would cope with whatever we had to get through as we would be guided and given strength to do so. We also, many of us, believed that after death, we went ‘back home’ to a better existence where we met again our loved ones, and this helped us cope with the grief of separation.
This is not the case any more. For many professional medics and others it would now be called ‘magical thinking’. I even hesitate to admit to it myself here, expecting some degree of derision, but in my own anxiety, which springs from uncertainty about the future, I find that trusting in some greater ‘universal force for good’ is what helps me to calm it down. I guess it’s the last taboo, the thing people don’t like to discuss. When I read Leonie’s post about meeting her son in a dream, I dared to admit that I had met mine that way too.
So, okay, this may be ‘magical thinking’ and ‘when we’re dead we’re dead’ etc. But maybe it isn’t….. We just don’t know. And maybe when stresses come, and we have problems dealing with the ensuing anxiety about how on earth we will cope, things like meditation to help us be calm and enable us to think without being overcome with panic, will help.
Whilst I’ve got my head above the parapet like this, I feel I’d like to admit something else. The whole reason for the work RxISK is doing, the difference it is making in highlighting the horrendous symptoms some people have been and are suffering for which they blamed themselves because no one told them otherwise, all this is really GOOD work, and maybe part of some much needed plan because this situation has reached crisis point worldwide.
By finding RxISK Katinka Newman understood what had happened to her, and hence she’s been able, with her book and website, to make the public more aware. She’s put up a film on her Blog now, made by a brave man called Josh who suffers from very severe akathisia, and he shows us what it is actually like. I realise, having seen it, that our son’s movements were just like this at times, but he was simply told he was anxious and attention-seeking, and given higher doses of various drugs. All doctors and health professionals should watch this and learn, and so should we all, to protect ourselves and our loved ones by making ourselves aware of these warning signs.
Good things done by good people inspire and enable more good things to happen. Tragedies happen, but out of those can come good things, and in the end, terrible powerful wrongs can be stopped.
Heather, thanks for your kind words – I didn’t mean to make out that I am in a bad way with the ‘anxiety’ because I am not. Suffice to say that from the days that our son was put on Seroxat until roughly 12 months ago, my days have been taken up in trying to get people to understand what went wrong when that prescription was given out, and why the problems were not helped by further prescriptions. Being able to fight on has kept me sane, no doubt about that ( well sane-ish anyway!).
Twelve months ago, a different psychiatrist started working with him, in a very different way, and my anxiety more or less disappeared. Last week, however, our son had a very bad week which meant a return to the old pattern of a trip to a&e etc. Immediately this happened I felt a terrible guilt for having been so laid back over the last twelve months – the old signs of the anxiety returned. Thankfully all is as well as expected again now, and I have been able to return to my relaxed mode!
I agree with what you say about ways of dealing with death etc. having changed over the years. I find it very interesting that, when there was a high percentage of belief in God/ everlasting life etc. people coped so much better with their ‘earthly life’ than the present day generations do. What there is, or isn’t, for us after our time on this earth is immaterial to my mind. If, by believing in our own little ‘heaven’ as a reward for our earthly trials, we are better able to cope with the present life then I can only see it as a good thing.
‘Everything you always wanted’ to know about Paroxetine and Adult Patients from those examining suicidality in adults with depression and other psychiatric disorders prior to approviing paroxetine..
Paroxetine and adult patients
Introduction
GlaxoSmithKline has been examining the issue of suicidality in adults with depression and other psychiatric disorders since before paroxetine was approved.
http://www.gsk.com/en-gb/media/resource-centre/paroxetine/paroxetine-and-adult-patients/
With Study329 and Continuation329 now published on RxISK and GSK filling up Alltrials Space, how Open is Open Trials…?
OpenTrials “search engine” for clinical trials nearing beta launch
The soon-to-be launched OpenTrials project aims to create an open, easy-to-use, linked database of information about the world’s clinical trials. It draws together information from multiple sources and presents the information in a variety of formats. The overall aim of the project is to improve access to information about trials, improve access to research, and boost transparency in the field. OpenTrials will launch a public beta on 10th October at the World Health Summit in Berlin, preceded by an OpenTrials Hack Day.
http://www.alltrials.net/news/opentrials-clinical-trials-transparency/
Can someone explain this post in plain, simple English? I’d really appreciate that..
Thanks…
Looks to me like the continuation phase didn’t produce a desirable outcome, even if they tweaked it, and seemed to result in negative information about the drug’s longer-term potential. In the absence of a positive outcome the study results were put away while the idea of long-term prescribing was promoted anyway. JAACAP, who published and will not retract the acute phase of the study (which sucked but researchers portrayed the drug positively), will not publish the continuation phase of the study submitted by the RIAT team (a study which should have been paid attention to by the original reseachers and this is why it needed to be published).
Thank you Laurie!
Is it ok if I post your explanation on my blog?
David Healy’s posts are brilliant, but sometimes can be too brilliant for my poor Seroxat damaged brain to digest..
🙂
Totally okay!
Totally agree Truthman. So glad you had the sense to say this. I tried reading it over and over again but my brain couldn’t cut to the chase either, and I don’t have the excuse of Seroxat, I think I am just not good at all this clever stuff – wish I was – but I’m working on it! If it’s any comfort, I noticed that it was quite some time before anyone put their toe in the water and made a comment. I bet we were all waiting for someone to say what you had the honesty to do! I know I was 🙂 so many thanks for that, and to Laurie too.
By Ed Silverman @Pharmalot
September 30, 2016
Glaxo to pay $20 million for bribing doctors in China
https://www.statnews.com/pharmalot/2016/09/30/glaxo-bribes-china-sec/
Antidepressant suicide warnings ‘backfired’
BBC News Report
with
U.S. Food and Drug Administration
Questions and Answers on Antidepressant Use in Children, Adolescents, and Adults:
Finally, the committee recommended that labeling remind health care professionals that depression and other disorders being treated with these drugs are serious illnesses that need to be recognized and effectively treated.
http://www.fda.gov/Drugs/DrugSafety/InformationbyDrugClass/ucm096321.htm
Date created: March 22, 2004, updated May 2, 2007
http://www.fda.gov/Drugs/DrugSafety/InformationbyDrugClass/ucm096305.htm
The proposed new Warning also alerts prescribers, patients, and their caregivers to the emergence of certain symptoms that are known to be associated with antidepressants in some patients, in particular, anxiety, agitation, panic attacks, insomnia, irritability, hostility, impulsivity, akathisia (severe restlessness), hypomania, and mania. Although a causal link has not been established between the emergence of such symptoms and either worsening of depression or the emergence of suicidal impulses, medications may need to be discontinued when such symptoms are severe, abrupt in onset, or were not part of the patient’s presenting symptoms.
Taken from:
Historical Information on Antidepressant Use in Children, Adolescents, and Adults
http://www.fda.gov/Drugs/DrugSafety/InformationbyDrugClass/ucm096293.htm
“Antidepressant suicide warnings ‘may have backfired’,” BBC News reports
Thursday June 19 2014
http://www.nhs.uk/news/2014/06June/Pages/Antidepressant-suicide-warning-backfired.aspx
https://www.madinamerica.com/2016/10/pills-steal-generations-lives/
A Chapter from Katinka on ‘New Look’ Mad in America..
https://www.madinamerica.com/
This is so crucial as to what happens when you might have stopped all the drugs but it never, ever leaves you as free as you were and it is probably impossible to have 0/10 plastered all over the pages where ‘authoratative’ persons got it all wrong……..
Being labelled mentally ill
http://www.thepillthatsteals.com/blog/
Anne-Marie 11 hours ago
This is very important, how do you correct this kind of medical mistake? because as you rightly said it can affect all kinds of things from jobs to mortgages e.t.c. I would be very interested to see what they say but I would imagine like many government agency’s they will just refer you back to the Dr who prescribed you the drug in the first place. Surely somehow you can have your records corrected. Could you ask the complaints dept of the hospital to find out what you can do about it? Someone, somewhere has to correct this injustice.
Well said..Anne-Marie
“I suppose you moved home to be nearer Kate” spat the gp at me as our relationship devolved..I was a guest in her house with my daughter on the Isle of Bute. She left us alone with her alcoholic partner as she travelled back to our village surgery to do two days work there.
Kate was the practice nurse on whom I became entirely dependant “if I were you Annie I would take all the pills available to me” she said. This was at the time of acute akathisia in her car off to our riding school, kids in the back of the car, terrified, as I was sobbing, garbling, holding on to Kate’s hand as she drove and I should have been in a hospital with people sorting out Paroxetine not sitting in someone’s car with a woman telling me to pop a bag full of anti-depressants, benzodiazepines and beta-blockers…
It all ended very badly.
I don’t move anywhere to be near anyone and the gp was so full of herself, so full of rightious indignation that she did the unforgiveable…..when I had left the mental hospital where she had sent me and three days later was in a general hospital with suicide from Seroxat, she did something so obscure, so ridiculous, so inept, that it is hard to see how this crazed woman’s mind worked…..she sat and went through almost the entire content of my medical records….she brought up medical history going back decades…to try and prove what a dipstick I was and, yet, all through the Seroxat disaster she did not ask me one personal question or make even one attempt to personalise herself with my life as it was.
She was not aware of my day to day life of art classes, all my village friendships, my daughters horse riding since age 6, my daughters scottish dancing achievements, my daughters scholarly achievements; my international airline pilot and his achievements in Africa, Antartica, Far East; my career in marketing and publicity in Canada and America and my role in promoting British Goods abroad.
All this passed her by as she put the knife in and savaged us and I honestly don’t know how I ever came out the other end…
Annie, the more I read about your misfortunes with the medical people, the more I feel that your story, just like Katinka’s, needs to be heard by as many people as possible. Katinka’s experience was a short sharp shock whereas yours seems to have been a long, drawn-out affair. Katinka came out the other end complete whereas you are still suffering today. Two differing stories under differing circumstances but both with the one, same warning – beware the many faces of “medical care”!
In three short extracts Dr. David Healy explains clearly much of the information we need to know and it needs to be put up again in case anyone missed it…
Dr. David Healy speaking at a Press Conference prior to the 2006 FDA Advisory Committee Public Hearing on the connection between antidepressants and suicidal thoughts and actions. The press conference was put together by the family of Woody Witczak.
https://truthman30.wordpress.com/2012/05/24/anti-depressant/
Appreciation to usual and reliable sources..
Annie shows us how we can all find ourselves judged by those professionals charged with our health care, who may not have the time to fully comprehend what has made us temporarily unwell, but who seem to feel they have the right to fit us into a mould which tallies with their personal opinions of us.
I guess this is a sort of ‘power’ thing. It’s not, I’m ashamed to say, only limited to doctors. I can remember when I worked as a social worker 40 years ago, we had clients who came to our Department and one social worker might write their opinion of their situation on their file, which could be, at best, inaccurate due to lack of listening time, and at worst, prejudiced because of personal issues with the client. Luckily, as we were in a department of several, and another social worker might meet the client at some later time, with more insight, the note taking could be altered. But I don’t think this was often the case. I was a very junior member of the team, tasked with making practical arrangements for all the patients. I had referrals to do this from all the senior social workers, so I met the clients too, in the course of so doing. It frankly horrified me to see the way very biased accounts were sometimes recorded, about people who were at their most vulnerable and in most need of being carefully listened to, and given the benefit of the doubt. I’m afraid it’s human nature when in positions of power over others, for some people to become blasé and their hubris gets the better of them. But we need to be well aware of this, and question judgements regularly.
Katinka draws clear attention to the horrendous consequences of being judged as mentally ill when in fact it was purely medications that caused her to behave oddly. To get those judgements off her record, she is now going to have to fight the authorities, even though none of this is her fault. The same thing happened to our son. Ridiculous opinions from a relative who was himself unreliable, were used to cement the diagnosis of a psychiatrist hard pressed for time, about an honest, trusting young man who was simply acting like Katinka, totally out of character, due to his inability to tolerate such drugs. I believe he and I and so many others like Annie who contribute to this Blog, are members of the 1% of people who cannot metabolise certain medications. Katinka says in her small and brilliant YouTube film this week ‘The Pill that Steals Lives’ that there are over 100 prescribed drugs on the market now which can affect the mind and body and trigger terrible effects, leading in some cases to suicide, as happened to our son. I prefer the more accurate term, coined by Kristina about her daughter Natalie (see Kidnapped post on RxIskblog), PRESCRIPTICIDE..
To change this situation, all patients should be allowed at the time to read what is being written about them, and sign it off, just as one would do when giving a statement to police. They should also have a totally independant advocate to read it with them, just to make sure the patient’s family have not put a biased slant on the situation, because once these entries are written down, it’s very difficult to alter opinions, and the baton can be passed from doctor to doctor. It can affect a person’s whole life, and even death.
On the point of incorrect medical records, I was most impressed last week, when our son came back from an appointment with his care co-ordinator and said that he’d been told that when he is off his medications, they will be asking the psychiatrist who deals with his reductions to re-assess and give his feelings re: diagnosis ” so that your records can be corrected”. I certainly hope that this is carried out, if and when such changes are appropriate.
One of the most disturbing films you will ever see in your life..Josh and akathisia and dystonia
http://www.thepillthatsteals.com/blog/
Aime and Josh”s lives have been torn apart by long term side effects of antidepressants and antipsychotics.
I watched Josh bravely showing his suffering from akathisia. My son was moving almost as continuously and in the same way in the weeks before he died, and even back in 2002/3 he was so unable to keep still after he took and later stopped Seroxat, that we got him a mini trampoline so that he didn’t keep running up and down the stairs. He even ran up and down the car park of our GPs surgery, but we and he were told either that it was anxiety, or later that it was attention seeking. Seeing Josh, it was just like seeing Olly again. I can’t believe the doctors really didn’t know it was akathisia; did they not say anything because they knew it was hopeless and there was nothing they could offer and they didn’t dare admit it?
Olly decided to die because he just could not understand what was happening to him. If the GP and psychiatrist had had the guts and the honesty to explain and reassure him, he might still have died, but I don’t think so, I think knowledge helps a lot and brings hope. I am more disgusted with the medics now than words can say. Poor Olly, and poor all sufferers at the hands of Big Pharma and all those who sustain them.
Heather – I do not know where you are taking this figure of 1% from, for adverse drug reactions. But it plays right into the hands of doctors & psychiatrists, in that they can claim that you are ‘one of the unlucky few’, or that it is own ‘your fault’ that you are ‘medication non-compliant’.
When my ex-partner was on a low dosage of an SSRI, it was definitely helping her. But when that dosage was doubled, she suffered from appalling akathisia and intense emotional blunting. In the grip of severe Stockholm Syndrome, she told me it was ‘all of her own fault’. Unfortunately, she’d bought into the medical BS big time…
I would suggest that you acquire a copy of Jay S Cohen’s book ‘Over Dose: The Case Against the Drug Companies’, written in 2001. All of these prescribed drugs are toxic poisons, and at high enough doses, EVERYONE will suffer an adverse reaction. But as we are all different, who dictates what might be a high or low dose for any particular individual?
Drug companies only wish to sell at a dosage that will ‘catch’ most people, as exemplified by the shenanigans which went into marketing Prozac. During the first Hindmarsh Zoloft trial at Leeds University in 1983, no less than 100% of the participants taking the active drug developed symptoms of akathisia, causing the trial to be prematurely terminated.
Genetic factors MAY have a relevance here, but until reliable tests are devised, we simply do not know.
I really wish this 1% quotation would just disappear! It does us no favours…
Walter
Walter, I got the figure from Katinka Newman’s YouTube videos on her ‘the Pill that Steals Lives’ site and I assumed that this was correct, so I do apologise if I’ve got this wrong but I quoted it in good faith. I guess I assumed this was an official figure as I think she used it in her interview on ITV.
I will certainly read Jay Cohen’s book, and thank you for the recommendation.
No need to apologise Heather. Concrete statistics within this iatrogenic world seem almost impossible to obtain – what we can be fairly certain about is that whatever ‘official’ figures are put forward about ADRs, the reality may exceed these numbers by a magnitude or several. Like many others on here, I too have followed Katinka’s story, and am pleased for her that the outcome was positive.
Your own story beggars belief, and no-one has a more compelling narrative, or a stronger voice, than a mother who has lost her child to these ghastly horrors. I hope that the passage of time will give you some sort of resolution to what you and your partner have been through.
Walter
Thanks Walter. I’ve ordered Dr Jay Cohen’s book ‘Overdose’ from the USA, and also read a a bit more about him. He sounds a marvellous guy. Interesting that he did a lot to champion those put on ‘flox’ antibiotics. My husband (aged 77) had an infection and GP proudly wrote out prescription for Cipro (a flox antibiotic). Aha, we have access to RxISK now and checked. Lots of horror stories re muscle/tendon damage, dementia, all kinds of side effects. In PIL it stipulates not to give it to anyone over 60! We agonised over weekend whilst he downed Vitamin C and cranberry juice. Dare we ring GP on Monday and ask for milder antibiotic, without upsetting him. Damage limitation conversation between us, husband said if his legs didn’t work and he could no longer do long walks, garden or sail, life over for him. So risked it and got less potent one. Losing GP would be better than losing joy in living 🙂 !
whaddayaknow
“Of all the antidepressants, Paxil was the only one to show a statistically significant association with suicide in depressed adults in the short and deeply flawed clinical trials used for FDA approval of the drug.”
David’s blog …
Continuation Phase …
Jury Awards $11.9 Million in Paxil Suicide Malpractice Case
http://www.bioportfolio.com/news/article/2863816/Jury-Awards-11-9-Million-in-Paxil-Suicide-Malpractice-Case-Psychiatric-Expert.html
I knew I had a Malpractice Case against a rural surgery and a small rural mental hospital which is why I had to almost beg and plead many law firms in Glasgow to take me on..
I had an interview with a Lady Lawyer where I had to explain my legal aid funded case in ten minutes. As I spoke, this lawyer tapped out everything I was saying on her computer and then gave me the Devastating News. “This Case will have to go to a GP, of my choosing, to back up your case.” At this point, I knew I was doomed..
I was.
The LegalGP did a copy and paste job of the Gps indiscretions and fabrications and a copy and paste job of the psychiatrists complete failure to assess my ‘medications’ during my week long stay in his mental hospital and who ‘forgot’ ‘failed’ ‘ignored’ his letter to the surgery when he had warned of Seroxat Discontinuation Symptoms occurring at cessation and what to do.
The letter had been stuffed in my medical records at the time of starting Seroxat and was only referred to by the lady gp in a complete fabrication when I was now coming down off 40 mg. Seroxat with the hospital pharmacist doing a year long plan with reduction of tablets and Paroxetine Liquid.
My dose of 40 mg. had been upped because of a violent attack on myself and ensuing personal trauma, but, I wanted off and I was getting off..
The gps fabrication about what was and what was not given to me when I was once again climbing the walls towards the end of the year long plan was completely evident when she ran in to my sitting room with the ‘advised drug’ which should have been given to me at the time of being taken off Seroxat and ‘not’ almost a year and a half later when I had been thrown into two hospital within three days of each other.
Rosemary was extremely sharp, a few years older than me, with labradors.
I had a labrador.
During this fateful year and a half, somehow I was walking her dogs as she couldn’t cope with being a gp, far from home on the I of B. The invitation to stay with her was before the ‘liquid drop’ but after the ‘violent attack’ and ’40 mg. Increase’.
A more clearer case of Stockholm Syndrome is unlikely than this.
The catalogue of errors by both psychiatrist and gp was a massive breach of not only ‘errors’ in prescribing but also the ‘bullying’ element which is obvious to anyone reading her three referrals but of course not to the psychiatrist who completely ignored me in his mental hospital.
Like Katinka was taken off all her drugs and had a month of terror and illness, but survived it, when the gp brought me the final drug as she knew by my behaviour that a second ‘terror’ attack was likely by me, all those anti-depressants, benzodiazepines and beta-blockers were binned and subsequently I was now cold turkey 5 drugs and to my eternal regret, we fled our life whilst I had to try and get better.
Having an 11 year old and the big licky lab probably saved me from too many years of profound drug abuse, as they were my focus and having to force myself to weed peoples gardens and clean their toilets to provide, from v nice people, probably also got me back on track and honed my mind to attempt to ignore my completely battered nervous system…
The Legal Eagle was completely useless and it was a sad day when the report came in from Glasgow suggesting I might be disappointed with the result..I read it once, that was enough..
It has to take an ‘Expert’ in matters of Psychotropic Drug Malpractice reading ‘through the lines’ in Medical Records to get to the heart of the ‘errors’ and it is a sad state of affairs that as the Law Stands Here in the UK, unless you can fund it yourself, a Law Firm won’t act for you and even then it is unlikely to succeed as who is to prove that Paroxetine leads to
violence/aggression/suicide/prescripticide/akathisia
My Formal Complaints to both entities, lasted years, ending with the complete desire to get rid of me with both entities taking the ‘bull by the horns’ and taking ‘their irritations’ to both NHS Highland and the Doctor and Dental Union of Scotland for corroboration ‘behind closed doors’ and that was the ‘end of the matter’..
I was told by a Barrister that gp’s are not experts. The only problem is then the local Psychiatrist will back them up not us. Being on an island must have made things even harder for you to deal with Annie, down here near London its still a problem but at least you can do more about it like change surgery’s and get a different gp e.t.c. I would imagine being on an island they would all stick together and leave you behind as the victim with no where to turn. Sounds like something from the “Wicker man”, doesn’t surprise me at all. Your strong and brave to stand up to them.
Sharing Peter *You’re evaluating junk!* Breggin for those not aware……and Alastair *the evidence, however, is clear, these medicines are not linked with suicide* Benbow
The evidence however is clear..
https://www.youtube.com/watch?v=sH4Kb41EoXc
The Evidence, However, Is Clear
https://www.amazon.co.uk/Evidence-However-Clear-Seroxat-Scandal/dp/1849914141/ref=cm_cr_arp_d_product_top?ie=UTF8
Our Comments..
https://www.amazon.co.uk/product-reviews/1849914141/ref=cm_cr_dp_see_all_summary?ie=UTF8&showViewpoints=1&sortBy=helpful
Fortitude is the marshal of thought, the armour of the will, and the fort of reason.
Francis Bacon
Product Liability
http://www.fortitudelaw.uk/product-liability/seroxat-group-action/
Patients were reassured by their GPs that unlike other antidepressants, they would be able to stop taking Seroxat whenever they wanted. Instead, over 6,000 individuals advised their GPs that each time they reduced their dose they suffered bizarre and debilitating symptoms not previously experienced. These symptoms often included impulsive suicidal thoughts, thoughts of self harm and uncharacteristic aggressive behavior. Their withdrawal symptoms were so severe that the only way they could be avoided was to return to their previous daily dose. Several of the current Claimants were minors at the time they were prescribed Seroxat for transient mental healthcare problems.
“It is ironic” says Claimant Bob Fiddaman “this British company has compensated Americans for the harm caused to adults and children who became dependent upon its antidepressant Paxil (Seroxat) yet UK citizens have had to fight for years to have their case heard. We may now only be a group of 105 Claimants but we are determined to gain compensation for the harm caused to us.”
I happened across a tweet on Alltrials giving a Live Stream on Friday with this Conference and, actually, it was really interesting.
I don’t normally spend a few hours listening to this sort of thing, but, I was in the mood for a bit of mental stimulation…..
At 4.27 Dr. Goldarca was introduced
At 4.54 Alltrials Campaign
At 4.55.30 we got on to GSK
“GSK signed up pretty early on and what is nice about that is..
10 days..
15 days..
Streamed live on 7 Oct 2016
The International Journal of Epidemiology editorial team invite you to a conference to celebrate the successes (and failures) of journal publication of epidemiological findings and the ways in which epidemiology has developed over the 44 years of the journal’s existence.
https://www.youtube.com/watch?v=WKiGCP1qiaU
Spot the deliberate mistake..
http://www.alltrials.net/news/roadmap/
This is a constantly evolving document and we would appreciate any feedback at alltrials@senseaboutscience.org
http://senseaboutscience.org/
Does your doctor wear a small and metal badge..