Editorial Note: This post is by Ken Spriggs, who wrote the second ever RxISK story – on Azathiopine. This should be revisited in the light of the AbbVie Campaign. The current post is taken from his ongoing blog on Crohn’s Disease – a condition that is high on the target list for the makers of biologicals including AbbVie and Humira.
Drug side effect ratios explained
This is the second part in a series of research articles focused on drugs discussed by IBD patients on crohnsforum.com. The concept of Drug Side Effect Ratio is defined.
The previous article took a look at what drugs IBD patients discuss on crohnsforum. This article examines the instances of the phrase “side effects” near the drug names. I collected Google results by using a site specific search then determined the percentage of mentions of “side effect” near the drug name.
Here’s an example, Google search: [“Remicade” “side effects” site:crohnsforum.com]. There were 6,250 results. From the previous article the total mentions of Remicade is 22,100. Dividing side effects results by total results we get 0.28. So the result is that 28% of the mentions of “Remicade” occur close to the phrase “side effects”. I’ll call this metric the Drug Side Effect Ratio. The higher the ratio the more we expect the discussions in the forum are focused on side effects.
I’ve done the analysis on a subset of the drugs used in the previous article. I’ve picked the top five common drug categories and only the drugs in each category which have the highest number of mentions. I also treated the drug category ‘biologic’ as a specific drug name. Generics are in lower case and trade names are capitalized.
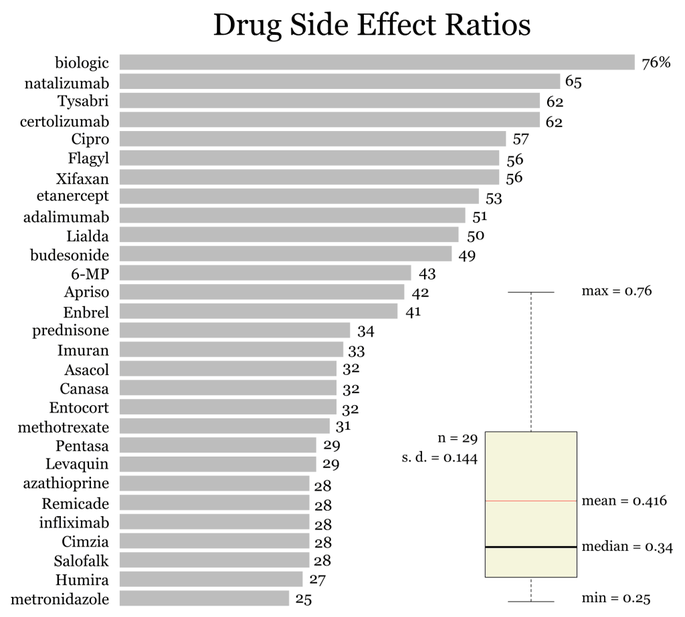
Let’s do a quick sanity check on the results. Notice that all the ratios are less than one. That’s good because if they weren’t it would mean there are more mentions of “side effects” than the drug name. Now consider the ranges of the distribution. The highest Drug Side Effect Ratio is for natalizumab which was withdrawn by the manufacturer and subsequently returned under the special prescription program. It’s not approved in the EU for Crohn’s treatment specifically because of side effects. Natalizumab’s trade name, Tysabri, has the second highest Drug Side Effect Ratio. The lowest Drug Side Effect Ratio is for metronidazole, but isn’t remarkably lower than the median. It’s trade name, Flagyl, on the other hand, is above the mean. It also seems reasonable to me that 76% of the discussion about biologics concerns itself with side effects.
Since biologics has the highest ratio let’s take a closer look at that category.
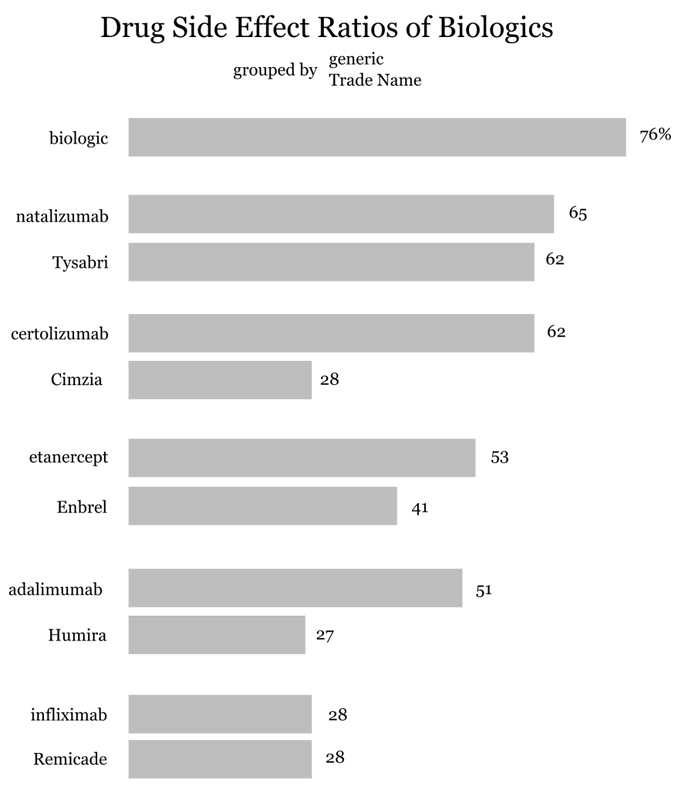
Drug side effect ratios for biologics
There’s a trend which seems to emerge. The trade names and generic names for the same drug have considerably different Drug Side Effect Ratios. Are people biased to use the generic name when discussing side effects? Does advertising play a role in the mentions of a trade name when mentioning side effects? It appears as if this is the trend when patients are discussing biologics. The observation doesn’t hold true for metronidazole (Drug Side Effect Ratio = 25%) and it’s trade name Flagyl (Drug Side Effect Ratio = 56%). What’s the context of discussion when a trade name is used versus a generic? Could sentiment analysis using natural language processing tell us anything about how patient dialogue differs regarding generic names and trade names?
More work is needed but it looks plausible that the Drug Side Effect Ratio could be useful in analyzing a patient message board like crohnsforum. For investigating the Drug Side Effect Ratio further a more rigorous parsing of the text is necessary. It’s possible the technique could be extended to using specific side effects near the drug names. For example the term fatigue could be counted near the drug name azathioprine.
Perhaps the results don’t say much in isolation but I feel like this analysis sheds light on what’s important in the IBD community. Drug Side Effect Ratio is certainly a step in considering descriptive generalized metrics about online patient communication.
This is a peculiar way of presenting frequency of side effects, just based on the number of mentionings. Is this adjusted for ad campaigns, or press releases/articles? Why do some drugs appear twice? e.g. flagyl and metronidazole? This is a bit like looking at who is the best fotball player by number of google mentionings: Beckham is number 3, but he is retired…
Not many comments lately, although, I am sure they come in thick and fast.
My hoover is broken. A trade name took over the entire vacuuming world.
If you extrapolate, as you have done, above, it will become apparent that most of the drugs you question will have a forum for it, a blog for it, a patient plead for information for it, that far surpasses the abilities of a gp and/or psychiatrist.
If you took this a stage further and extrapolated all hits on all sites, then you would need a team of folk to do the statistics.
I am sure you have a job to do, as well as write books and do everything in your power to highlight dictats from pharma, dictats from gps/psychiatrists and dictats from government agencies ie MHRA, NICE, EMA, and so all I am saying is would all blogs on the dangers of ssris, in particular, send in their hit list to Professor Healy.
Is that reasonable, so that an informed statistical analysis can take place…….. I am quite sure you are far too busy to take on the entire google search for all the drugs that have questionable safety records, but, then, I am not Professor Healy, who seems to bound through and have an energy, without, which, a lot would be lost on the general public and general practitioners and qualified psychiatrists.
All it takes is a questioning mind.
So, far, so good. What’s next after extrapolations. Could it be ‘just a minute syndrome’. I really thought I had no problems with my medication, but, just a minute, it seems I do.
Over to you, to do your job, Professor Healy, and doing it, you are…….
This week my GP said he could prescribe me an antidepressant or refer me to a rheumatologist. I could tell he was leaning towards the Rx when he said ‘Antidepressants are safe’. Unbelievable.
Not sure if he was going with a brand or a generic because I asked for the referral.
This is some really cool work – and it’s not the first time that “data-mining” of Internet chat has been used to pick up evidence of drug problems that aren’t being reported to (or investigated by) official bodies. Back in March some Stanford researchers announced they’d picked up a previously unknown drug interaction problem by data-mining patient discussion sites:
http://www.nytimes.com/2013/03/07/science/unreported-side-effects-of-drugs-found-using-internet-data-study-finds.html?_r=1&
There’s a team at West Virginia U. that has high hopes of doing the same:
http://online.wsj.com/article/SB10000872396390443982904578044440117124454.html
It’s interesting to think of how these data-miners could collaborate with a more focused effort like RxISK.org. By fanning out to scan billions of messages, they could detect lots of “signals” that indicate a problem but are not high-quality enough to tell you anything about cause and effect. The RxISK database is much, much smaller, but gathers lots of information about the how and why of people’s drug experiences, and even allows people to be re-contacted if they wish to be. Working together, these two types of projects might find out a lot.
As for why the generic names turn up more when people complain about side effects? Sometimes the only patients who even know the generic names of their drugs are those who have started to question their doctors, because they’ve had a problem. Maybe just knowing the word “adalimumab” and being able to spell it is enough to classify you as a Squeaky Wheel — one of those awful patients who Goes On The Internet?
There is a huge focus on adherence with drugs even when they are hard to tolerate.
Across so many disease ‘profiles’. Is it possible for patients who must take pill regimens for life , to have the level of drug measured over several doses to see if there is an over prescribing of their meds – metabolism differs , time to clear doses between one and the next ..differ from person to person. I have wanted to do this with my regimen for a long time as if I am honest with Doctor ( rotating , changes every app. no continuity) unless I adjust , I cannot tolerate for longer than 3 days without huge difficulty even after a year . But the pharma company wants ‘efficiency’. If I don’t I am ‘failing to adhere’. I am the failure. I am the problem. I am not the client patient , I am the subject patient – a liability , if I do not do as the guideline directs. There is no inbetween ground to negotiate with although silently and regularly patients do this to try to be ‘adherent’ without the Doctor every knowing . But this patient wisdom is not tapped into . It is as if we are breaking a law if we modify a guideline and intuit for ourselves what is a safe limit with what are basically poisons.
The man is doing a great job. Remember, he started out in psychiatry, and you can’t blame him for his disgust with where that’s going. New diagnoses are flying left and right – from ADHD to bipolar and a new “defiant disorder” (Note they mainly target children, the most vulnerable group). That’s how Behavioural therapy got its start, first slowly with animals, then to discipline children, then BT with phobias and limited mental ailments, then graduated to Cognitive-Behavioural or CBT to include the use of cognitive skills for people to convince themselves to behave better), and so on. Meanwhile, the DSM gets ever fatter! There is no doubt its fraud, but who’s to blame? It’s just a vacuum that’s being filled by shrewd pharma, ready and eager to make money.
To get anywhere with innovative or non-mainstream ideas you have to be very socially proactive – nay, aggressive. Personally, I have a wonderful, highly effective form of psychotherapy that is particularly effective with children and often “cures” their aberrant behaviours in record time, which I’ve been trying to impart for 20 years, but I’m outnumbered. It’s like trying to get people to change their religion. So I can appreciate how daunting the task is for Healy. I say lets keep an open mind and give this guy some credit.