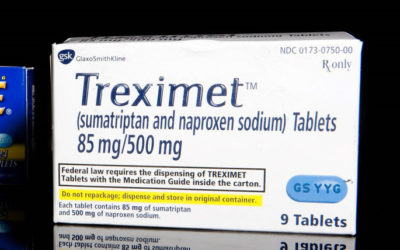
Study 329 Trick, Treat or Treximet
The plan this morning was to continue coverage of the Opioid Epidemic but perhaps because it is Halloween a link to a Wall Street Journal story on how Drug Makers Turn Cheap Generics into Expensive Pills arrived by email. It was difficult to resist. Here’s why. The article features Treximet, a combination of sumatriptan and naproxen, … Continue reading Study 329 Trick, Treat or Treximet
39 Comments